Please refer to the Hydrogen Revision Notes given below. These revision notes have been designed as per the latest NCERT, CBSE and KVS books issued for the current academic year. Students will be able to understand the entire chapter in your class 11th Chemistry book. We have provided chapter wise Notes for Class 11 Chemistry as per the latest examination pattern.
Revision Notes Chapter 9 Hydrogen
Students of Class 11 Chemistry will be able to revise the entire chapter and also learn all important concepts based on the topic wise notes given below. Our best teachers for Grade 11 have prepared these to help you get better marks in upcoming examinations. These revision notes cover all important topics given in this chapter.
Position of Hydrogen in Periodic Table :
♦ Lightest element known having atomic number 1.
♦ Dihydrogen
♦ It resembles both alkali metals and halogens and therefore, its position is anomalous.
♦ In modern periodic table it is located separately
Resemblance with alkali metals:-
1. Electronic configuration
1H = 1s1 11Na = 1s2, 2s2, 2p6, 3s1 19K = 1s2, 2s2, 2p6, 3s23p6, 4s1
2. Electropositive character: H+, Na+, K+ etc.
3. Oxidation state: +1
4. Combination with electronegative elements: form binary compounds with electronegative elements like alkali metals.
Halides: HClNaCl, KCletc
Sulphides: H2S Na2S, K2S etc
Resemblance with halogens:-
1. Electronic configuration: Both contain one electron less than the nearest noble gas configuration
1H = 1s1 (near to 2He)
9F = 1s2, 2s2, 2p5 (near to 8Ne)
17K = 1s2, 2s2, 2p6, 3s23p5 (near to 18Ar)
2. Non-metallic character: like halogens, hydrogen is non-metallic in nature.
3. Atomicity: Diatomic molecules.
4. Formation of similar types of compounds:
i. Halides: CCl4, SiCl4, GeCl4
ii. Hydrides: Cl4, SiH4, GeH4
5. Oxidation state: –1
Na+1H-1 Na+1Cl-1
Difference from alkali metals:-
1. Ionization enthalpy: the ionization enthalpy of hydrogen is very high in comparison to alkali metals.
2. Non- metallic character: alkali metals are typical metals while hydrogen is non-metal
3. Atomicity: hydrogen is diatomic while alkali metals are monoatomic.
4. Nature of compounds: the compounds of hydrogen are predominantly covalent while those of alkali metals are ionic. For example: HCl is covalent while NaCl is ionic.
The oxides of alkali metals are basic while hydrogen oxide is neutral.
Difference from halogens:-
1) Less tendency for hydride formation:Hydrogen has less tendency to take up electron to form hydride ion (H-) as compared to the halogens which from halide ions (X-) very easily.
2) Absence of unshared pairs of electrons :
3) Nature of oxides: The oxides of halogens are acidic while hydrogen oxide is neutral.
Occurrence of Hydrogen:
Hydrogen, the most abundant element in the universe and the third most abundant on the surface of the globe, is being visualised as the major future source of energy
Isotopes of hydrogen:

Preparation:
Methods for commercial production of dihydrogen
1. Electrolysis of water
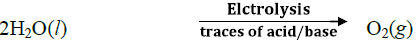
The hydrogen prepared by this method is of very high purity. However, this method is not commonly used because it is very expensive. This method is can be used only at those places where the electricity is cheap.
2. By the reaction of steam on coke :
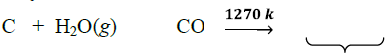
♦ Since the mixture of CO and H2 is used for the synthesis of methanol and a number of hydrocarbons, it is also called synthesis gas or syn gas.
♦ The process of producing syn gas from coal or coke is called coal gasification.

♦ This reaction is called water gas shift reaction.
Properties of Hydrogen:
Physical Properties:
1) It is slightly soluble in water (about 2 %)
2) It is highly combustible and therefore should be handled carefully.
3) It lightest substance. The weight of one litre hydrogen at NTP is only 0.0899 g.
Chemical properties: Not very reactive due to high bond dissociation energy (435.88 kJ mol-1 at 298.2 K)
(i)Combustion: It burns with pale blue flame
2H2 (g) + O2(g) ⟶ 2H2O(l)
(ii) Reaction with metals: Reactive metals like Na, K, Ca, Li and form hydrides
Ca + H2CaH2⟶525 k
Metals like Pt, Pd, Ni (elements of d block) form interstitial hydrides by absorbing large volume of hydrogen. Such hydrogen is called ‘occluded hydrogen and this property of adsorption of a gas by a metal is called occlusion.
(iii) Reaction with metal oxides: Hydrogen reduces oxides of less active metals to corresponding metal
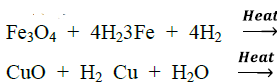
(iv) Reaction with non-metals:
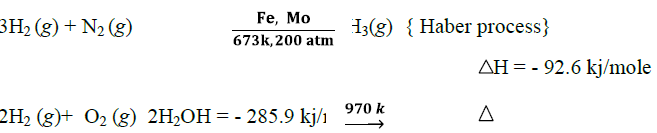
(v) Reaction with carbon monoxide:

(b)Hydroformylation of olefins to aldehydes:Hydroformilation or Oxo process
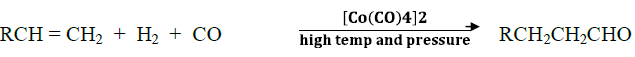
(c) Hydrogenation of oils:Vegetable oils are polyun-saturated in nature. The C =C bonds in oils can easily undergo oxidation and the oil becomes rancid i.e.,
unpleasant in taste. Hydrogenation reduces the number of double bonds but completely.
Vegetable Oil + H2 Ni/473 k ⟶ (Solid)
Uses of Hydrogen:
1. as a reducing agent.
2. In the manufacture of vanaspati fat, ammonia, metal hydrides, methanol, fertilizers such as urea etc.
3. In the manufacture of synthetic petrol.
4. In the atomic hydrogen torch and oxy hydrogen torches for cutting and welding. Dihydrogen is dissociated with the help of an electric arc and the hydrogen atoms produced are allowed to recombine on the surface to be welded. High temperature of about 4000 k is generated.
5. In the fuel cell for generating electrical energy.
Ortho and parahydrogens:-A molecules of dihydrogen …….abc
♦ They show different physical properties. For example :
(i) The thermal conductivity of para hydrogen is about 50 % greater than that of ortho hydrogen.
(ii) The melting point of para hydrogen is 0.15 k below that of hydrogen containing 75% ortho hydrogen.
♦ They show similar chemical properties.
Atomic hydrogen:
♦ Because of high H—H bond enthalpy, atomic hydrogen is produced only at high temp in an electric arc or under ultraviolet radiation.

♦ Highly reactive.
♦ Half life period is 0.3 sec and therefore, it immediately gets converted into the molecular form liberating a large amount of energy which is used for cutting and welding purposes
Nascent hydrogen:The hydrogen produced in contact with the substance to be reduced is known as ‘nascent hydrogen ’. It is very reactive form of hydrogen Better reducing agent than ordinary dihydrogen.
Hydrides: Under certain conditions H2 combines with almost all the elements ,except noble gases to form compounds called hydrides.
♦ There are three types of hydrides ,they are
(i) Ionic or saline hydrides: These are the compounds of H2 formed with most of the s-block elements which are highly electro positive.
(ii) Covalent or molecular hydrides: These are the compounds of hydrogen formed with most of the p-block elements
Electron deficient: The hydrides which do not have sufficient number of electrons to form normal covalent bonds is called electron deficient hydride. For example, hydride of group 13 (BH3, AlH3, etc.).They are known as Lewis acids i.e., electron acceptors. To make up their deficiency they generally exist in polymeric forms such as B2H6, Al2H6, etc.
Electron precise: The hydrides which have sufficient number of electrons required for forming covalent bonds is called electron precise hydride. For example, hydrides of group 14 (CH4, SiH4, GeH4, SnH4, PbH4 etc.) they have tetrahedral geometry.
Electron rich hydrides: The hydrides which have excess electrons as required to form normal covalent bonds is called electron rich hydride. For example, hydrides of group 15 to 17 (NH3, PH3, H2O, H2S, H2Se, H2Te, HF etc.)
(iii) Metallic or non-stoichiometric hydrides:
♦ These are formed by many d-block and f-block elements
♦ These hydrides conducts heat and electricity though not efficient.
Water: Water! It is the major part of all living organisms.water is also known as the river of life.
Human body has about 65%and some plants haveasmuch as 95%water.
Structure of Water :
In a gas phase water is bent molecule with a bond angle of 104.5 and O-H bond length of 95.7pm It is highly polar molecule.

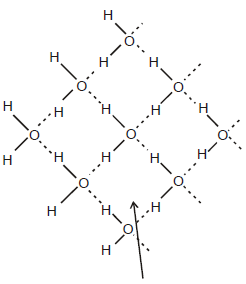
Structure of Ice :
Ice has a highly ordered 3D hydrogen bonded structure. Each oxygen atom is surrounded tetrahedrally by four other four other oxygen atoms at a distance of 276 pm
Chemical Properties of water:
AMPHOTERIC NATURE:It has the ability to act as an acid as well as base .it acts as an acid with NH3 and as a base with H2S
H2O(l) +NH3(aq)⟶ OH–(aq) +NH4+ (aq)
H2O(l) +H2S(aq) ⟶ H3O+(aq) +HS– (aq)
REDOX REACTIONS INVOLVING WATER: Water can be easily reduced to H2 by highly electropositive metals
2H2O(l) +2Na(s) ⟶ 2NaOH(aq) +H2(g)
HYDROLYSIS REACTION:Due to high dielectric constant, it has a very strong hydrating tendency .it dissolves many ionic compounds
P4O10(s) +6H2O(l) ⟶ 4H3PO4(aq)
SiCl4(l) +2H2O(l) ⟶ SiO2(s) + 4HCl(aq)
HYDRATES FORMATION: From the aqueous solutions many salts can be crystallized as hydrated salts. It of different types.
(1) Coordinated water e.g., [Cr(H2O)6]3+ 3Cl–
(2) Interstitial water e.g.,BaCl2.2H2O
(3) hydrogen-bonded water e.g. [Cu(H2O)4]2+4SO2- .H2Oin CuSO4.5H2O
Hard & Soft Water:
♦ The water which contains dissolved salts of bicarbonates, sulphates and chlorides of calcium and magnesium is called hard water. Hard water does not produce lather with soap solution.
♦ Soft water is free from bicarbonates, sulphates and chlorides of calcium and magnesium. It produces lather with soap solution easily. e.g., distilled water, rain water.
Types of hardness:The hardness of water is of two types
(i)Temporary hardness
♦ Due to presence of soluble bicarbonates of calcium and magnesium.
♦ Can be removed by simple boiling.
(ii)Permanent hardness
♦ Due to presence of chlorides and sulphates of calcium and magnesium.
♦ Requires treatment of water to remove this type hardness.
♦ Do you know?
Temporary hardness is also called carbonate hardness &
Permanent hardness is also called non-carbonate hardness
Softening of water: The process of removal of Ca2+ and Mg2+ ions from water is called softening of water.
Removal of temporary hardness:
(i) By boiling :
heat
M(HCO3)2 → MCO3 + H2O + CO2(M = Ca or Mg)
(Soluble) (Insoluble)
(ii) Clark’s method or calcium hydroxide method :
Ca(HCO3)2 + Ca(OH)2 →2CaCO3 + 2H2O
(Soluble) (Insoluble)
Mg(HCO3)2 + 2Ca(OH)2 → 2CaCO3 + Mg(OH)2 + 2H2
(Soluble) (Insoluble)(Insoluble)
Removal of permanent hardness:
(i) By washing soda (Na2CO3.10H2O) treatment:
CaCl2 +Na2CO3→ 2CaCO3 + 2NaCl
(Insoluble)
MgSO4 +Na2CO3 → MgCO3 + Na2SO4
(Insoluble)
(ii) By using inorganic cation exchanger (permutit method or Zeolite method):
Na2Al2Si2O8 + CaCl2→Ca(Al2Si2O8)2 + 2NaCl
Zeolite Settles at bottom
The zeolite can be regenerated by treatment with sodium chloride solution.
Ca(Al2Si2O8)2+ 2NaCl → Na2Al2Si2O8 + CaCl2
(iii) By organic ion exchanger or synthetic resins (ion exchange resins):
♦ Synthetic resins are the insoluble polymeric solids having giant hydrocarbon network containing reactive acidic or basic groups. These are superior to Zeolitebecause they can remove all types of cations as well as anions present in water. This resulting water is known as demineralised or deionised water.
♦ These are two types: H+
(a) Cation exchanger resins: they have acidic groups such as COOH or SO3H. they may be represented as resin—H+
Mg2+ + 2H-resin ⟶ Mg (resin)2 + 2H+
In hard water Cation exchanger
Ca2+ + 2H- resin ⟶ Ca(resin)2 + 2H+
Hard water Cation exchanger
(b) Anion exchanger resins: they have basic groups such as –OH– or –NH2.they may be represented as resin—OH– or resin—NH3 +OH–
SO42 + 2HO – resin ⟶ SO4resin2 + 2OH–
Hard water Anion exchanger
Cl– + HO-resin ⟶ Cl-resin + OH–
Hard water Anion exchanger
Regeneration of resiners:
Cl – resin + NaOH ⟶ HO – resin + NaclH–
Exchausted resin Re generated resin
Hydrogen peroxide [H2O2]: discovered by French chemist J.L. Thenard
Methods of preparation
1. From sodium peroxide (Merck’s process):
Na2O2 + H2SO4 → Na2SO4 + H2O2
(20% ice cooled solution) (30% solution)
2. From Barium peroxide:
a. Hydrogen peroxide was first prepared by J. L. Thenard in 1818 by acidifying barium peroxide and removal of excess water by evaporation under reduced pressure.
BaO2.8H2O + H2SO4 ⟶ BaSO4+ 8H2O + H2O2
b. Barium sulphate is filtered off leaving behind H2O2.
Store of Hydrogen peroxide:
a) Itmust be kept in wax lined coloured bottles because the rough glass surface causes its decomposition.
b) A small amount of phosphoric acid , glycerol or acetanilide is generally added which retard the decomposition of H2O2. These are also called negative catalysts.
Physical properties of Hydrogen peroxide:
1. In the pure state H2O2 is an almost colourless(very pale blue) liquid.
2. H2O2is miscible with water in allproportions and forms a hydrate H2O2.H2O(mp 221K).
3. A 30% solution of H2O2 is marketedas ‘100 volume’ hydrogen peroxide. It means thatone millilitre of 30% H2O2solution will give 100 Vof oxygen at STP. Commercially, it is marketedas 10 V, which means it contains 3% H2O2.
Chemical properties of Hydrogen peroxide:
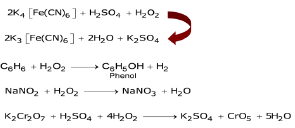
♦ Oxidising properties
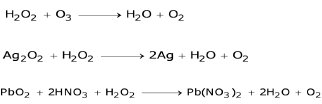
♦ Reducing properties
♦ Uses of hydrogen peroxide
1) For bleaching silk, wool, hair and leather
2) As rocket fuel
Structure of hydrogen peroxide

Hydrogen economy (Hydrogen as fuel)
♦The electricity cannot be stored to run automobiles. It is not possible to store and transport nuclear energy. Hydrogen is an alternative source of energy and hence called as ‘hydrogen economy’. Hydrogen has some advantages as fuel
♦ Available in abundance in combined form as water.
♦ On combustion produces H2O. Hence pollution free.
♦ H2-O2 fuel cell give more power.
♦ Excellent reducing agent. Therefore can be used as substitute of carbon in reduction for processes in industry.
Obstacles in hydrogen economy
♦ Transportation:
♦ Hydrogen gas is explosive and hence it is difficult to store and transport.
♦ Formation of hydrogen from H2O:
♦ The cheaper production of the hydrogen is basic requirement of hydrogen economy which is not possible now.
♦ The main aim and advantage of hydrogen economy is to transmit energy in four of hydrogen.


