Please see Respiration in Plants Class 11 Biology Revision Notes provided below. These revision notes have been prepared as per the latest syllabus and books for Class 11 Biology issues by CBSE, NCERT, and KVS. Students should revise these notes for Chapter 14 Respiration in Plants daily and also prior to examinations for understanding all topics and to get better marks in exams. We have provided Class 11 Biology Notes for all chapters on our website.
Chapter 14 Respiration in Plants Class 11 Biology Revision Notes
♦ Oxidation of food materials (breaking of C-C bonds of complex molecules) within the cell to release energy for ATP synthesis is called cellular respiration.
♦ This energy is used for absorption, transport, movement, reproduction, breathing etc.
♦ Ultimate source of food that is respired is photosynthesis.
♦ The compounds that are oxidized during respiration are called respiratory substrates. E.g. Carbohydrates (most common), proteins, fats and organic acids.
♦ The energy released is not used directly but is used to synthesize ATP. When energy is needed, ATP is broken down. Hence, ATP acts as energy currency of the cell.
BREATHING IN PLANTS
♦ For respiration, plants get O2 and give out CO2.
♦ In plants, gas exchange occurs via stomata & lenticels.
♦ Plants need no specialized respiratory organs because
1. Each plant part takes care of its own gas-exchange needs. So gas transport is very limited.
2. Very low gas exchange as compared to that of animals.
3. Leaves are adapted for maximum gas exchange during photosynthesis. During this, O2 is released within the cell.
4. Most living cells have contact with air. They are located close to plant surface. In stems, living cells are organized in thin layers beneath the bark. They also have lenticels. In leaves, stems & roots, parenchyma cells are loosely packed that provides interconnected air spaces.
♦ Complete combustion of glucose yields energy most of which is given out as heat.
C6H12O6 + 6O2 → 6CO2 + 6H2O + Energy
♦ This energy is utilized to synthesize other molecules.
♦ During the glucose catabolism, not all the liberated energy goes out as heat. Glucose is oxidised in several small steps. It enables some steps to couple released energy to ATP synthesis .
♦ During respiration, oxygen is utilized, and CO2 , water & energy are released.
♦ Certain organisms are adapted to anaerobic conditions. Some are facultative anaerobes. Others are obligate.
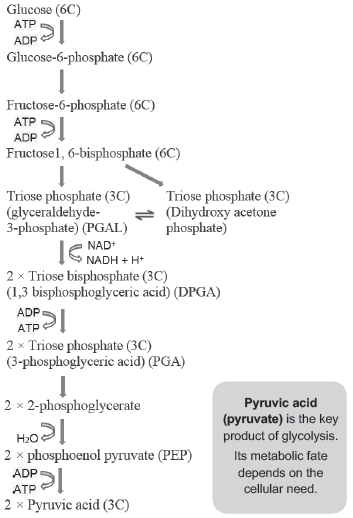
GLYCOLYSIS (EMP PATHWAY)
♦ It is the partial oxidation (breakdown) of glucose to 2 molecules of pyruvic acid (C3H4O3) in the absence of O2.
♦ It occurs in cytoplasm of all living organisms.
♦ Its scheme was given by Gustav Embden, Otto Meyerhof & J. Parnas. So it is also known as EMP pathway.
♦ In anaerobes, it is the only process in respiration.
♦ In plants, glucose is derived from sucrose (end product of photosynthesis) or from storage carbohydrates. Sucrose is converted to glucose & fructose by an enzyme, invertase. These 2 monosaccharides readily enter glycolytic pathway.
♦ Glucose & fructose are phosphorylated to form glucose-6- phosphate by the enzyme hexokinase. It is then isomerised to produce fructose-6-phosphate. Subsequent steps of metabolism of glucose and fructose are same.
Steps of glycolysis:
♦ It includes 10 steps under the control of different enzymes.
♦ ATP is utilized at 2 steps:
1. In the conversion of glucose into glucose 6-phosphate.
2. In the conversion of fructose 6-phosphate to fructose 1, 6-diphosphate.
♦ Fructose 1, 6-diphosphate is split into dihydroxyacetone phosphate (DHAP) & 3-phosphoglyceraldehyde (PGAL).
♦ PGAL is oxidised and with inorganic phosphate get converted to 1, 3-bisphosphoglycerate (BPGA). During this, 2 redox-equivalents (2 H-atoms) are removed from PGAL and transferred to NAD+ forming NADH + H+.
♦ BPGA becomes 3-phosphoglyceric acid (PGA) yielding energy. This energy is trapped by the formation of ATP.
♦ ATP is also formed when PEP converts to pyruvic acid.
♦ In glycolysis, 4 ATP molecules are directly synthesised from one glucose molecule.
In different cells, pyruvic acid is handled in 3 ways:
♦ Lactic acid fermentation.
♦ Alcoholic fermentation.
♦ Aerobic respiration (Krebs’ cycle).
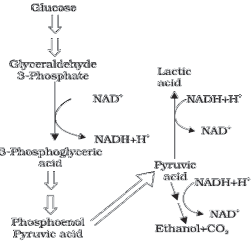
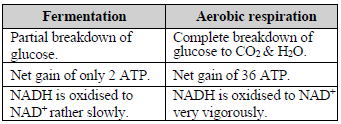
FERMENTATION (ANAEROBIC RESPIRATION)
♦ It is the incomplete oxidation of glucose under anaerobic condition.
♦ It occurs in many prokaryotes and unicellular eukaryotes.
♦ It is 2 types:
Alcoholic fermentation: Here, the pyruvic acid formed from glucose is converted to CO2 and ethanol. The enzymes, pyruvic acid decarboxylase and alcohol dehydrogenase catalyse these reactions. E.g. Yeast. Yeasts poison themselves to death when the concentration of alcohol reaches about 13%.
Lactic acid fermentation: Here, pyruvic acid is converted to lactic acid. E.g. Some bacteria.
♦ The reducing agent (NADH+H+) is reoxidised to NAD+ in both the processes.
♦ In animals, when oxygen is inadequate during exercise, pyruvic acid in muscle cells is reduced to lactic acid by lactate dehydrogenase.
♦ Net ATP production from fermentation of one glucose molecule = 2. (4 ATP from glycolysis – 2 ATP utilized).
♦ The steps involved in fermentation are shown below:
Drawbacks of fermentation
♦ Energy production is limited. Less than 7% of the energy in glucose is released and not all of it is trapped as high energy bonds of ATP.
♦ Hazardous products (acid or alcohol) are formed.
AEROBIC RESPIRATION
♦ It is a complete oxidation of organic substances in the presence of oxygen releasing CO2, water & energy.
♦ It occurs in mitochondria.
♦ For this, the pyruvate (final product of glycolysis) is transported from the cytoplasm into the mitochondria.
♦ The crucial events in aerobic respiration are:
1.Complete oxidation of pyruvate by stepwise removal of all the hydrogen atoms, leaving 3 CO2 molecules. It takes place in the matrix of mitochondria.
2. Passing on of electrons removed as part of H-atoms to molecular O2 with simultaneous synthesis of ATP. It occurs on the inner membrane of mitochondria.
♦ Pyruvate (pyruvic acid) enters mitochondrial matrix and undergoes oxidative decarboxylation in presence pyruvic dehydrogenase. It needs coenzymes, NAD+ & Coenzyme A.
♦ During this process, 2 NADH molecules are produced from 2 pyruvic acid molecules
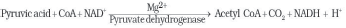
♦ Acetyl CoA then enters tricarboxylic acid (TCA) cycle.
Tricarboxylic Acid Cycle
(Krebs’ cycle or Citric acid cycle)
TCA cycle was first elucidated by Hans Krebs.
Steps:
1. Condensation of acetyl group with oxaloacetic acid (OAA) & water to form citric acid in presence of citrate synthase enzyme. A CoA molecule is released.
2. Citrate is isomerised to isocitrate.
3. Decarboxylation of isocitrate to α-ketoglutaric acid.
4. Decarboxylation of α-ketoglutaric acid to succinyl-CoA.
5. Succinyl-CoA is converted to succinic acid and a GTP molecule is synthesised (substrate level phosphorylation). In a coupled reaction, GTP is converted to GDP with simultaneous synthesis of ATP from ADP.
6. Oxidation of succinate to Fumarate and then to Malate.
7. Oxidation of malate to OAA.
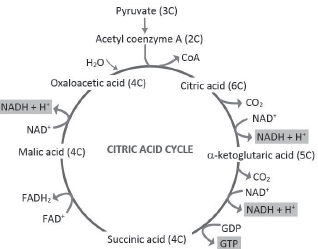
♦ At 3 points of TCA cycle, NAD+ is reduced to NADH + H. At one point, FAD+ is reduced to FADH2.
♦ Continued oxidation of acetyl CoA via TCA cycle requires continued replenishment of OAA. It also requires regeneration of NAD+ & FAD+ from NADH & FADH2.
Summary equation of Krebs’ cycle:
mitochondrial Matrix
Pyruvic acid+ 4NAD++FAD++2H2O +ADP+ PI → 3CO2 + 4NADH +4H++ FADH2 + ATP
Thus, a glucose is broken down to give 6 CO2, 8 NADH+H+, 2 FADH2 and 2 ATP.
Electron Transport System (ETS) & Oxidative Phosphorylation
♦ Electron transport system (ETS) is the metabolic pathway present in the inner mitochondrial membrane through which electron passes from one carrier to another.
♦ This is to release and utilize energy stored in NADH+H+ nd FADH2 (formed during TCA cycle) by oxidation.
♦ The electrons are passed on to O2 to form H2O.
♦ Electrons from NADH are oxidised by an NADH dehydrogenase (complex I).
♦ Electrons are then transferred to ubiquinone (UQ) located within the inner membrane. Ubiquinone also receives reducing equivalents via FADH2 (complex II) that is generated during oxidation of succinate in citric acid cycle.
♦ The reduced ubiquinone (ubiquinol or UQH2) is then oxidised with the transfer of electrons to cytochrome c via cytochrome bc1 complex (complex III). Cytochrome c is a small protein attached to the outer surface of the inner membrane. It acts as a mobile carrier of electrons between complex III and IV.
♦ Complex IV (cytochrome c oxidase) contains cytochromes a & a3, and 2 copper centres.
♦ When the electrons pass from one carrier to another via complex I to IV, they are coupled to ATP synthase (complex V) for the ATP production.
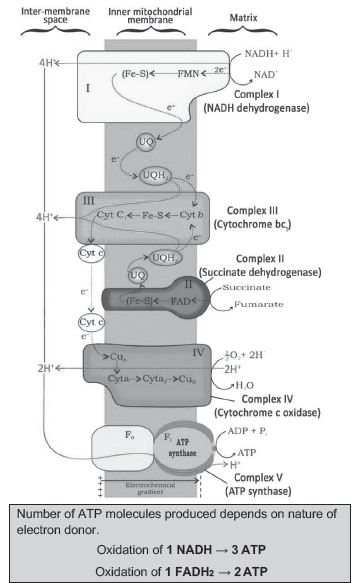
♦ In aerobic respiration, the role of oxygen is limited to the terminal stage. Yet, oxygen is vital since it drives the whole process by removing hydrogen from the system. Oxygen acts as the final hydrogen acceptor.
♦ In respiration, energy of oxidation-reduction is utilised for the phosphorylation. So this process is called oxidative phosphorylation. It is not as photophosphorylation (Here, light energy is utilised to produce proton gradient for phosphorylation).
♦ The energy released during the ETS is utilized to synthesize ATP by ATP synthase (complex V).
♦ ATP synthase has two major components: F1 & F0.
♦ F1 headpiece (peripheral membrane protein complex): Site for ATP synthesis from ADP & inorganic phosphate.
♦ F0 (integral membrane protein complex): It forms a channel through which protons cross the inner membrane. The passage of protons is coupled to the catalytic site of the F1 component for ATP production.
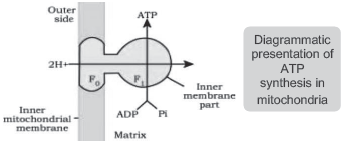
♦ For each ATP produced, 2H+ passes through F0 from the inter-membrane space to the matrix down the electrochemical proton gradient.
THE RESPIRATORY BALANCE SHEET
♦ Net gain of ATP from each glucose molecule is calculated based on the following assumptions:
1. All steps in Glycolysis, TCA cycle & ETS occur sequentially and orderly.
2. The NADH synthesised in glycolysis is transferred into mitochondria and undergoes oxidative phosphorylation.
3. Intermediates in the pathway are not used to synthesise other compounds.
4. Only glucose is being respired. Other alternative substrates are not entered in the pathway at any stages.
♦ Such assumptions are not valid because,
1. All pathways work simultaneously and do not take place one after another.
2. Substrates enter the pathways and are withdrawn from it as and when necessary.
3. ATP is utilized as and when needed.
4. Enzymatic rates are controlled by multiple means.
♦ Such calculations are useful to appreciate the efficiency of the living system in extraction and storing energy.
Net gain of ATP molecules from one glucose molecule
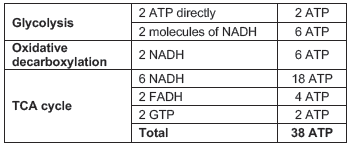
2 ATP molecules are spent for transporting 2 NADH molecules formed during glycolysis to the mitochondria. Hence the net gain = 36 ATP molecules.
Comparison b/w fermentation & aerobic respiration
AMPHIBOLIC PATHWAY
♦ Glucose is the favoured substrate for respiration. So, all carbohydrates are first converted to glucose for respiration.
♦ Other substrates are also respired.
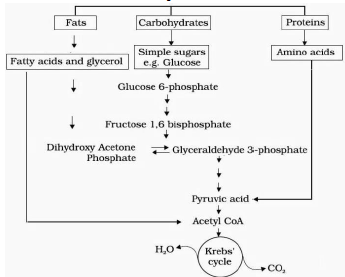
♦ Fats breakdown into glycerol & fatty acids. Fatty acids are degraded to acetyl CoA and enter the pathway. Glycerol is converted to PGAL and enters the pathway.
♦ Proteins are degraded by proteases into amino acids. Each amino acid (after deamination) enters the pathway at some stage in the Krebs’ cycle or as pyruvate or acetyl CoA.
♦ The respiratory pathway is generally considered as a catabolic pathway. But it involves both anabolism (synthesis) and catabolism (breakdown). So it is better called as an amphibolic pathway.
E.g. Fatty acids breakdown to acetyl CoA before entering the respiratory pathway. But when the organism needs to synthesise fatty acids, acetyl CoA withdraw from the respiratory pathway. Similarly, during breakdown and synthesis of protein, respiratory intermediates are involved.
RESPIRATORY QUOTIENT (RQ) OR RESPIRATORY RATIO
♦ It is the ratio of the volume of CO2 evolved to the volume of O2 consumed in respiration.
Volume of CO2 evolved
RQ = ________________________
Volume of O2 consumed
♦ RQ depends upon the type of respiratory substrate.
♦ RQ for carbohydrates= 1, because equal amounts of CO2 and O2 are evolved and consumed, respectively.
C6H12O6 + 6O2 → 6CO2 + 6 H2O + energy
6 CO2
RQ =________ = 1.0
6O2
♦ RQ for fats = < 1. Calculations for a fatty acid, (e.g.tripalmitin) are shown:
2 (C51H98O6) + 145O2 → 102 CO2 + 98 H2O + energy
102CO2
RQ = ________ = 0.7
145O2
♦ RQ for proteins = 0.9.
♦ In living organisms, respiratory substances are often more than one. Pure proteins or fats are never used as respiratory substrates.



