Please refer to the Thermodynamics Revision Notes given below. These revision notes have been designed as per the latest NCERT, CBSE and KVS books issued for the current academic year. Students will be able to understand the entire chapter in your class 11th Chemistry book. We have provided chapter wise Notes for Class 11 Chemistry as per the latest examination pattern.
Revision Notes Chapter 6 Thermodynamics
Students of Class 11 Chemistry will be able to revise the entire chapter and also learn all important concepts based on the topic wise notes given below. Our best teachers for Grade 11 have prepared these to help you get better marks in upcoming examinations. These revision notes cover all important topics given in this chapter.
Thermodynamics: Science which deals with study of different forms of energy and quantitative relationship.
System & Surroundings: The part of universe for study is called system and remaining portion is surroundings.
State of system & state function: State of system is described in terms of T, P, V etc. The property which depends only on state of system not upon path is called state function eg. P, V, T, E, H, S etc.
Extensive & Intensive Properties: Properties which depends on quantity of matter called extensive prop. eg. mass, volume, heat capacity, enthalpy, entropy etc. The properties which do not depends on matter present depends upon nature of substance called Intensive properties. eg. T,P, density, refractive index, viscosity, bp, pH, mole fraction etc.
Internal energy: The total energy with a system.
i.e. U = Ee + En + Ec + Ep + Ek + ——
ΔU = U2 – U1 or UP – UR & U is state function and extensive properly. If U1 > U2 energy is released.
Heat (q): It I a form of energy which is exchanged between system and surrounding due to difference of temperature. Unit is Joule (J) or Calorie (1 Calorie = 4.18μ J).
First Law of Thermodynamics: It is law of conservation energy. Energy can neither be created not destroyed, it may be converted from one from into another.
Mathematically ΔU = q + w, w = –p.ΔV (work of expansion)
ΔU = q – p. ΔV or q = ΔU + p.ΔV, q,w are not state function.
But ΔU is state function.
Enthalpy (H): At constant volume ΔV = 0, qv = ΔU
So H = U + p.ΔV, qp = H2H1 = ΔH
⇒ΔH = ΔU + P.ΔV.
Relationship between qp, qv i.e. ΔH& ΔU
It is ΔH = ΔU+ Δng.RT or qp= qv + ng.RT
Exothermic and Endothermic reactions: ΔH = –Ve for exothermic and ΔH = +Ve for endothermic reaction i.e. evolution and absorption of heat.
Eg C+O2 CO2 + 393.5 KJ, ΔH = –393.5 KJ (exothermic)
N2 + O2 2NO – 180.7 KJ, ΔH = 180.7 KJ (Endothermic)
Enthalpy of reaction ( ΔrH): The amount of heat evolved or absorbed when the reaction is completed.
Standard Enthalpy of reaction (ΔrH°) at 1 bar pressure and specific temp. (290K) i.e. standard state.
Different types of Enthalpies of reactions:
(i) Enthalpy of combustion (ΔcH),
(ii) Enthalpy of formation (ΔfH)
(iii) Enthalpy of neutralization
(iv) Enthalpy of solution
(v) Enthalpy of atomization(ΔaH),
(vi) Enthalpy of Ionisation ( ΔiH)
(vii) Enthalpy of Hydration ( Δhyol.H)
(viii) Enthalpy of fusion ( Δfus.H)
(ix) Enthalpy of vaporization ( Δvap.H)
(x) Enthalpy of sublimation ( Δsub.H)
(Δsub.H) = ( Δfus.H) – ( Δvap.H)
Hess’s Law of constant heat summation: The total amount of heat change is same whether the reaction takes place in one step or in several steps.
i.e. ΔH = ΔH1 +ΔH2 + ΔH3 + ——–
Bond enthalpy: It is amount of energy released when gaseous atoms combines to form one mole of bonds between them or heat absorbed when one mole of bonds between them are broken to give free gaseous atoms. Further ΔrH= ∑B.E. (Reactants) – ∑B.E. (Products)
Spontaneous & Non Spontaneous Processes: A process which can take place by itself is called spontaneous process. A process which can neither take place by itself or by initiation is called non Spontaneous.
Driving forces for spontaneous process:
(i) Tendency for minimum energy state.
(ii) Tendency for maximum randomness.
Entropy (S): It is measure of randomness or disorder of system. i.e. Gas>Liquid>Solid.
Entropy change (ΔS) = q(rev.) / T J.K-1 .mol-1
Spontaneity in term of (ΔS)
ΔS(total) = ΔS(universe) = ΔS(system) +ΔS(surrounding)
If ΔS(total) is +ve, the process is spontaneous.
If ΔS(total) is –ve, the process is non spontaneous.
Second Law of thermodynamics: In any spontaneous process, the entropy of the universe always increases. A spontaneous process cannot be reversed.
Gibb’s free energy (G): defined as G = H – T.S & ΔG = ΔH – T. ΔS (Gibb’s Helmholts equation) it is equal useful work i.e. – ΔG = W(useful) = W(max.) If ΔG = ve, process is spontaneous.
Effects of T on spontaneity of a process: ΔG = ΔH – T. ΔS.
(i) For endothermic process may be non spontaneous at law temp.
(ii) For exothermic process may be non spontaneous at high temp. and spontaneous at law temp.
Calculation of (ΔrG°)
ΔrG°= ∑ΔfG° (p) – ∑ΔfG° (r)
Relationship between ( ΔrG°) & equilibrium constant (k)
ΔG = ΔG°+ RTlnQ & ΔG°= –2.303RT logk.
Calculation of entropy change:
ΔrS°= ∑ΔS°(p) – ∑S°(r)
Important Points
1. System and the Surroundings: A system in thermodynamics refers to that part of universe in which observations are made and remaining universe constitutes the surroundings. The surroundings include everything other than the system System and the surroundings together constitute the universe. The wall that separates the system from the surroundings is called boundary
2. Types of the System:
• Open System: In an open system, there is exchange of energy and matter between system and surroundings.
• Closed System In a closed system, there is no exchange of matter, but exchange of energy is possible between system and the surroundings.
• Isolated system :In an isolated system, there is no exchange of energy or matter between the system and the surroundings
3. State of a System: The state of a system means the condition of the system which is described in terms of certain observable properties such as temp(T), pressure(p), volume (v), etc. of the systems. These properties of a system are called state variables.
4. State Functions: A physical quantity is solid to be state functions of its value depends only upon the state of the system and does not depend upon the path by which this state has been attained.
5. Internal Energy: a quantity which represents the total energy of the system. It may be chemical, electrical and mechanical or any other type of energy you may think of, the sum of all these is the energy of the system. In thermodynamics, we call it the internal energy, U of the system.
6. Isothermal Process: When a process is carried out in such a manner that the temp remains constant throughout this process, it is called an isothermal process.
7. Adiabatic Process: Process is carried out in such a manner that no heat can flow from the system to the surrounding or vice versa.
8. Isochoric Process: Process during which the volume of the system is kept constant.
9. Isobaric Process: Process during which the pressure of the system is kept constant. .
10. The positive sign expresses that Work (wad) is positive when work is done on the system. Similarly, if the work is done by the system,wad will be negative.
11. The q is positive, when heat is transferred from the surroundings to the system and q is negative when heat is transferred from system to the surroundings.
12. First law of Thermodynamics: Statement: Energy can neither be created nor destroyed, however it may be converted from one form to another.
or
The total energy of the universe remains constant although it may undergo transformation from one to another.
Mathematical expression = Δ U = q + w
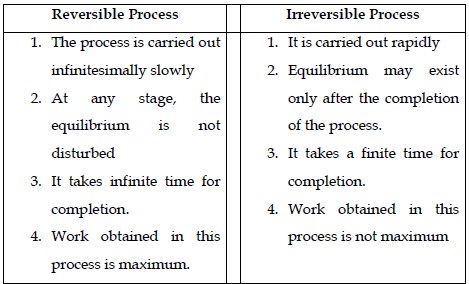
13. A process or change is said to be reversible, if a change is brought out in such a way that the process could, at any moment, be reversed by an infinitesimal change. A reversible process proceeds infinitely slowly by a series of equilibrium states such that system and the surroundings are always in near equilibrium with each other. Processes other than reversible processes are known as irreversible processes.
14. Difference between Reversible Process & Irreversible Process
15. Enthalpy, H: The enthalpy H [Greek word enthalpien, heat content] is defined as : H = U + pV For finite changes at constant pressure, we can write above equation as ΔH = ΔU + ΔpV Since p is constant, we can write ΔH = ΔU + pΔV since pΔV=ΔngRT ,therefore ΔH = ΔU + ΔngRT
16.Extensive property: An extensive property is a property whose value depends on the quantity or size of matter present in the system. For example, mass, volume, internal energy, enthalpy, heat capacity, etc. are extensive properties.
17. Intensive property: Those properties which do not depend on the quantity or size of matter present are known as intensive properties. For example temperature, density, pressure etc. are intensive properties.
18. Specific heat capacity is the quantity of heat required to raise the temperature of one unit mass of a substance by one degree celsius (or one kelvin). For finding out the heat, q, required to raise the temperatures of a sample, we multiply the specific heat of the substance, c, by the mass m, and temperatures change, ΔT as q=c×m×ΔT =CΔT .
19. Relationship between Cp and CV for an ideal gas: Cp -CV=R
20.Bomb calorimeter: For chemical reactions, heat absorbed at constant volume, is measured in a bomb calorimeter . Here, a steel vessel (the bomb)is immersed in a water bath. The whole device is called calorimeter. The steel vessel is immersed in water bath to ensure that no heat is lost to the surroundings. A combustible substance is burnt in pure dioxygen supplied in the steel bomb. Heat evolved during the reaction is transferred to the water around the bomb and its temperature is monitored. Since the bomb calorimeter is sealed, its volume does not change i.e., the energy changes associated with reactions are measured at constant volume. Under these conditions, no work is done as the reaction is carried out at constant volume in the bomb calorimeter. Even for reactions involving gases, there is no work done as ΔV = 0. Temperature change of the calorimeter produced by the completed reaction is then converted to qV, by using the known heat capacity of the calorimeter with the help of equation (18).
21. Enthalpy Change of a reaction: The enthalpy change accompanying a reaction is called the reaction enthalpy. The enthalpy change of a chemical reaction is given by the symbol ΔrH.
ΔrH = (sum of enthalpies of products) – (sum of enthalpies of reactants).
ΔrH=Σai HProducts– Σbi HReactants
22.The standard enthalpy of reaction is the enthalpy change for a reaction when all the participating substances are in their standard states. The standard state of a substance at a specified temperature is its pure form at1 bar. Standard conditions are denoted by adding the superscript o to the
symbol ΔH, e.g., ΔH°
23. The enthalpy change that accompanies melting of one mole of a solid substance in standard state is called standard enthalpy of fusion or molar enthalpy of fusion, Δfus.H°.e.g. H2O(s)→H2O(l); Δfus.H° =6.00 kJ mol−1.
24.Amount of heat required to vaporize one mole of a liquid at constant temperature and under standard pressure (1bar) is called its standard enthalpy of vaporization or molar enthalpy of vaporization, Δvap.H°. e.g.H2O(l)→H2O(g);Δvap.H°=40.79 kJ mol−1
25. Standard enthalpy of sublimation, ΔsubH°subis the change in enthalpy when one mole of a solid substance sublimes at a constant temperature and under standard pressure (1bar). Sublimation is direct conversion of a solid into its vapour. Solid CO2 (dry ice) sublimes at 195K with ΔsubH°=25.2 kJ mol−1; naphthalene sublimes slowly and for this ΔsubH0=73.0 kJ mol−1
26.The standard enthalpy change for the formation of one mole of a compound from its elements in their most stable states of aggregation (also known as reference states) is called Standard Molar Enthalpy of Formation. Its symbol is ΔfH°.
27. .Standard enthalpy of combustion is defined as the enthalpy change per mole (or per unit amount) of a substance, when it undergoes combustion and all the reactants and products being in their standard states at the specified temperature.
28. Enthalpy of atomization (symbol: ΔaH°): It is the enthalpy change on breaking one mole of bonds completely to obtain atoms in the gas phase. In case of diatomic molecules, like dihydrogen the enthalpy of atomization is also the bond dissociation enthalpy. In some cases, the enthalpy of atomization is same as the enthalpy of sublimation.
29. Bond Enthalpy (symbol: ΔbondH°):
ΔrH=Σbondenthapiesreactants– Σ bondenthapiesproducts
30. Enthalpy of Solution (symbol : ΔsolH° ): Enthalpy of solution of a substance is the enthalpy change when one mole of it dissolves in a specified amount of solvent. The enthalpy of solution at infinite dilution is the enthalpy change observed on dissolving the substance in an infinite amount of solvent when the interactions between the ions (or solute molecules) are negligible.
31. Lattice Enthalpy The lattice enthalpy of an ionic compound is the enthalpy change which occurs when one mole of an ionic compound dissociates into its ions in gaseous state.
32. Hess’s Law: Enthalpy change for a reaction is the same whether it occurs in one step or in a series of steps. This may be stated as follows in the form of: If a reaction takes place in several steps then its standard reaction enthalpy is the sum of the standard enthalpies of the intermediate reactions into which the overall reaction may be divided at the same temperature.ΔrH°
=ΔrH1+ΔrH2+ ΔrH3 For example: C(s) +
1/2O2 (g)→CO(g) ΔrH° =-110.5 kJmol−1 CO(g) + 1/2O2
(g)→CO2(g) ΔrH° =-283.0 kJmol−1 C(s) + O2
(g)→CO2 (g) ΔrH° = -393.5 kJmol−1
33. Spontaneous Process: A process which occurs by its ones i.e. Without the intervention of an outside agency.
34. Non Spontaneous Process: A process which can neither take place by itself nor by initiation is called a non spontaneous process.
35. Driving Force: The force which is responsible for spontaneity of a process is called the driving force.
36. Entropy(S): Entropy is a measure of randomness or disorder of the system.
37. Entropy change (ΔS): It is defined as the amount of heat (q) observed isothermally and reversibly divided by the absolute temp(T) at which the heat is absorbed.
ΔS = q / T
38. Entropy of fusion : ΔS fusion = ΔH fusion / Tm
Where, ΔH fusion = Entropy of fusion per mole
Tm = Melting point
ΔSVAP = ΔHVAP / Tb
ΔHVAP = Entropy of vaporization per mole
Tb =Boiling point
39. Second Law Of Thermodynamics : The entropy of universe is continuously increasing due to spontaneous process taking place in it.
ΔS system + ΔS surrounding > 0
i.e. ΔS total > 0
40. Gibbs Free Energy (G) : It is defined as maxm amount of energy available to a system during the process that can be converted into useful work. It is a measure of capacity to do useful work.
G = H – TS this equation is also known as Gibb‘s Helmholtz equation
G is a state function. ΔG is change in free energy.
ΔG = ΔH – TΔS
41. If ΔG is negative, process is spontaneous when ΔG =0,the process is in equilibrium if ΔG is positive, the process does not take place.
42. Standard Free Energy Change (ΔG° ) : It is defined as free energy change measured at 298 K and 1 atm Pressure.
43. Standard Free energy of formation: ( ΔfG° ) It is free energy change when 1 mole of compound is formed from its constituting elements in their standard state.
ΔrG° =∑vp ΔfG°(products) – ∑vr ΔfG°(reactants)
44. Gibbs Energy Change and Work
In case of galvanic cell, Gibbs energy change ΔrG ,is related to the electrical work done by the cell.
Thus ΔrG = -nFEcell
If reactants and products are in their standard states
ΔrG° = -nFE°cell, Here E°cell is the standard cell potential.
45. Gibbs Energy Change and Equilibrium Constant
ΔrG°= RT ln K
ΔrG°= – 2.303RT log K
Important Formulas
• Δ U = q + w
• ΔH = ΔU + pΔV
• ΔH = ΔU + ΔngRT
• Cp -Cv=R
• ΔrH=Σai HProducts– Σbi HReactants
• ΔrH=Σbondenthapiesreactants– Σ bondenthapiesproducts
• ΔrH° =ΔrH1+ΔrH2+ ΔrH3
• ΔS fusion = ΔH fusion / Tm
• ΔG =ΔH – TΔS
• ΔrG°= ∑vpΔfG°(products) – ∑vRΔfG°(reactants)
• ΔrG= -nFEcell
• ΔrG = -2.303RT log K

