Please refer to MCQ Questions Chapter 11 Dual Nature of Radiation and Matter Class 12 Physics with answers provided below. These multiple-choice questions have been developed based on the latest NCERT book for class 12 Physics issued for the current academic year. We have provided MCQ Questions for Class 12 Physics for all chapters on our website. Students should learn the objective based questions for Chapter 11 Dual Nature of Radiation and Matter in Class 12 Physics provided below to get more marks in exams.
Chapter 11 Dual Nature of Radiation and Matter MCQ Questions
Please refer to the following Chapter 11 Dual Nature of Radiation and Matter MCQ Questions Class 12 Physics with solutions for all important topics in the chapter.
MCQ Questions Answers for Chapter 11 Dual Nature of Radiation and Matter Class 12 Physics
Question. Silver has a work function of 4.7 eV. When ultraviolet light of wavelength 100 nm is incident on it a potential of 7.7 V is required to stop the photoelectrons from reaching the collector plate. How much potential will be required to stop photoelectrons, when light of wavelength 200 nm is incident on it?
(a) 15.4 V
(b) 2.35 V
(c) 3.85 V
(d) 1.5 V
Answer
D
Question. When a certain metallic surface is illuminated with monochromatic light of wavelength l, the stopping potential for photoelectric current is 3V0. When the same surface is illuminated with a light of wavelength 2λ, the stopping potential is V0. The threshold wavelength for this surface for photoelectric effect is
(a) 4λ
(b) 6λ
(c) 8
(d) (4/3)λ
Answer
A
Question. If the distance of 100 W lamp is increased from a photocell, the saturation current i in the photocell varies with the distance d as
(a) i ∝ d2
(b) i ∝ d
(c) t ∝ 1/d
(d) t ∝ 1/d2
Answer
D
Question. The following process is known as hu→ e+ + e−
(a) pair production
(b) photoelectric effect
(c) Compton effect
(d) Zeeman effect
Answer
A
Question. A metallic surface ejects electrons when exposed to green light of intensity I but no photoelectrons are emitted when exposed to yellow light of same intensity I. It is possible to eject electrons from the same surface by
(a) violet light of intensity more than I
(b) yellow light of any intensity
(c) red light of any intensity
(d) none of these
Answer
A
Question. The kinetic energy of an electron get tripled, then the de Broglie wavelength associated with it changes by a factor
(a) 1/3
(b) √3
(c) 1/√3
(d) 3
Answer
C
Question. An eye can detect 5 × 104 photons per square metre per second of green light (λ = 5000 Å) while the ear can detect 10–13 W m–2. The factor by which the eye is more sensitive as a power detector than the ear is close to
(a) 5
(b) 10
(c) 106
(d) 15
Answer
A
Question. In photoelectric emission process from a metal of work function 1.8 eV, the kinetic energy of most energetic electrons is 0.5 eV. The corresponding stopping potential is
(a) 1.3 V
(b) 0.5 V
(c) 2.3 V
(d) 1.8 V
Answer
B
Question. A and B are two metals with threshold frequencies 1.8 × 1014 Hz and 2.2 × 1014 Hz. Two identical photons of energy 0.825 eV each are incident on them. Then photoelectrons are emitted by
(Take h = 6.6 × 10–34 J s)
(a) B alone (b) A alone
(c) neither A nor B
(d) both A and B
Answer
B
Question. Light of two different frequencies whose photons have energies 1 eV and 2.5 eV, respectively, successively illuminate a metal whose work function is 0.5 eV. The ratio of the maximum speed of the emitted electrons will be
(a) 1 : 5
(b) 1 : 4
(c) 1 : 2
(d) 1 : 1
Answer
C
Question. The threshold frequency for a photosensitive metal is 3.3 × 1014 Hz. If light of frequency 8.2 × 1014 Hz is incident on this metal, the cut-off voltage for the photoelectric emission is nearly
(a) 2 V
(b) 3 V
(c) 5 V
(d) 1 V
Answer
A
Question. The frequency of incident light falling on a photosensitive metal plate is doubled, the kinetic energy of the emitted photoelectrons is
(a) double the earlier value
(b) unchanged
(c) more than doubled
(d) less than doubled
Answer
C
Question. An electron accelerated under a potential difference V volt has a certain wavelength l. Mass of proton is some 2000 times of the mass of the electron. If the proton has to have the same wavelength l, then it will have to be accelerated under a potential difference of
(a) V volt
(b) 2000 V volt
(c) (V/2000) volt
(d) √2000 V volt
Answer
C
Question. The maximum velocity of electrons emitted from a metal surface is v, when frequency of light falling on it is f. The maximum velocity
when frequency becomes 4f is
(a) 2v
(b) > 2v
(c) < 2v
(d) between 2v and 4v
Answer
B
Question. The ratio of momentum of an electron and a-particle which are accelerated from rest by a potential difference of 100 V is

Answer
D
Question. Light of wavelength l strikes a photosensitive surface and electrons are ejected with kinetic energy E. If the kinetic energy is to be increased to 2E, the wavelength must be changed to λ′ where

Answer
C
Question. The collector plate in an experiment on photoelectric effect is kept vertically above the emitter plate. Light source is put on and a saturation photo-current is recorded. An electric field is switched on which has a vertically downward direction, then
(a) the photo-current will increase
(b) the kinetic energy of the electrons will increase
(c) the stopping potential will decrease
(d) the threshold wavelength will increase
Answer
B
Question. The work function of a certain metal is 3.31 × 10–19 J. Then, the maximum kinetic energy of photoelectrons emitted by incident radiation of wavelength 5000 Å is (Given, h = 6.62 × 10–34 J s, c = 3 × 108 m s–1, e = 1.6 × 10–19 C)
(a) 2.48 eV
(b) 0.41 eV
(c) 2.07 eV
(d) 0.82 eV
Answer
B
Question. Two particles X and Y having equal charges, after being accelerated through the same potential difference, enter a region of uniform magnetic field and describe circular paths of radii R1 and R2, respectively. The ratio of masses of X and Y is
(a) (R1/R2)
(b) (R2/R1)
(c) (R1/R2)2
(d) (R2/R1)2
Answer
C
Question. Which phenomenon best supports the theory that matter has a wave nature?
(a) Electron momentum
(b) Electron diffraction
(c) Photon momentum
(d) Photon diffraction.
Answer
B
Question. A photon of energy E ejects a photoelectron from a metal surface whose work function is W0. If this electron enters into a uniform magnetic field of induction B in a direction perpendicular to the field and describes a circular path of radius r, then the radius r is given by, (in the usual notation)

Answer
D
Question. The velocity of a particle at which the kinetic energy is equal to its rest energy is


Answer
D
Question. A photo-sensitive material would emit electrons, if excited by photons beyond a threshold. To overcome the threshold, one would increase the
(a) voltage applied to the light source
(b) intensity of light
(c) wavelength of light
(d) frequency of light
Answer
D
Question. One electron and one proton is accelerated by equal potential. Ratio in their de-Broglie wavelength is
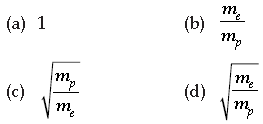
Answer
C
Question. A light whose frequency is equal to 6 × 1014 Hz is incident on a metal whose work function is 2 eV. [h = 6.63 × 10–34 Js, 1 eV = 1.6 × 10–19 J] The maximum energy of the electrons emitted will be
(a) 2.49 eV
(b) 4.49 eV
(c) 0.49 eV
(d) 5.49 eV.
Answer
C
Question. An electron microscope is used to probe the atomic arrangements to a resolution of 5 Å. What should be the electric potential to which the electrons need to be accelerated?
(a) 2.5 V
(b) 6 V
(c) 2.5 kV
(d) 6 kV.
Answer
B
Question. Two electrons are moving in opposite direction with speeds 0.8 c and 0.4 c, where c is the speed of light in vacuum. Then the relative speed is about
(a) 0.4 c
(b) 0.8 c
(c) 0.9 c
(d) 1.2 c
Answer
C
Question. The value of Planck’s constant is
(a) 6.63 × 10–34 J/sec
(b) 6.63 × 10–34 kg m2/sec
(c) 6.63 × 10–34 kg m2
(d) 6.63 × 10–34 J sec
Answer
D
Question. The wavelength of a 1 keV photon is 1.24 × 10–9 m. What is the frequency of 1 MeV photon?
(a) 1.24 × 1015
(b) 2.4 × 1020
(c) 1.24 × 1018
(d) 2.4 × 1023
Answer
B
Question. Monochromatic light of wavelength 667 nm is produced by a helium neon laser. The power emitted is 9 mW. The number of photons arriving per second on the average at a target irradiated by this beam is
(a) 3 × 1016
(b) 9 × 1015
(c) 3 × 1019
(d) 9 × 1017
Answer
A
Question. If a photon has velocity c and frequency u, then which of the following represents its wavelength?
(a) hu/c2
(b) hu
(c) hc/E
(d) hu/c
Answer
C
Question. By photoelectric effect, Einstein proved
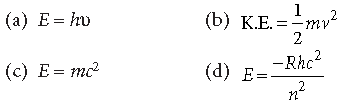
Answer
A
Question. Momentum of photon of wavelength λ is
(a) hu/c
(b) zero
(c) hλ/c2
(d) hλ/c
Answer
A
Question. The velocity of photons is proportional to (where V = frequency)
(a) 1/√V
(b) V2
(c) V
(d) √V
Answer
C
Question. A radio transmitter operates at a frequency 880 kHz and a power of 10 kW. The number of photons emitted per second is
(a) 1.72 × 1031
(b) 1.327 × 1025
(c) 1.327 × 1037
(d) 1.327 × 1045
Answer
A
Question. An electron of mass m and a photon have same energy E. The ratio of de-Broglie wavelengths associated with them is
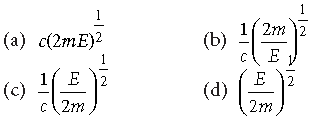
Answer
C
Question. Light of wavelength 500 nm is incident on a metal with work function 2.28 eV. The de Broglie wavelength of the emitted electron is
(a) ≥ 2.8 × 10–9 m
(b) ≤ 2.8 × 10–12 m
(c) < 2.8 × 10–10 m
(d) < 2.8 × 10–9 m
Answer
A
Question. The momentum of a photon of an electromagnetic radiation is 3.3 ×10–29 kg m s–1. What is the frequency of the associated waves? [h = 6.6 × 10–34 J s ; c = 3 × 108 m s–1]
(a) 1.5 × 1013 Hz
(b) 7.5 × 1012 Hz
(c) 6 × 103 Hz
(d) 3 × 103 Hz
Answer
A
Question. Monochromatic light of frequency 6.0 × 1014 Hz is produced by a laser. The power emitted is 2 × 10–3 W. The number of photons emitted, on the average, by the source per second is
(a) 5 × 1016
(b) 5 × 1017
(c) 5 × 1014
(d) 5 × 1015
Answer
D
Question. The momentum of a photon of energy 1 MeV in kg m/s will be
(a) 5 × 10–22
(b) 0.33 × 106
(c) 7 × 10–24
(d) 10–22
Answer
A
Question. The energy of a photon of wavelength λ is
(a) hcλ
(b) hc/λ
(c) λ/hc
(d) λh/c
Answer
D
Question. An electron is accelerated from rest through a potential difference of V volt. If the de Broglie wavelength of the electron is 1.227 × 10–2 nm, the potential difference is
(a) 10 V
(b) 102 V
(c) 103 V
(d) 104 V
Answer
D
Question. The de-Broglie wavelength of a neutron in thermal equilibrium with heavy water at a temperature T (kelvin) and mass m, is

Answer
A
Question. Electrons of mass m with de-Broglie wavelength l fall on the target in an X-ray tube. The cutoff wavelength (λ0) of the emitted X-ray is

Answer
A
Question. Which of the following figures represent the variation of particle momentum and the associated de-Broglie wavelength?

Answer
D
Question. An electron is accelerated through a potential difference of 10,000 V. Its de Broglie wavelength is, (nearly) (me = 9 × 10–31 kg)
(a) 12.2 nm
(b) 12.2 × 10–13 m
(c) 12.2 × 10–12 m
(d) 12.2 × 10–14 m
Answer
C
Question. A particle of mass 1 mg has the same wavelength as an electron moving with a velocity of 3 × 106 m s–1. The velocity of the particle is
(a) 3 × 10–31 ms–1
(b) 2.7 × 10–21 ms–1
(c) 2.7 × 10–18 ms–1
(d) 9 × 10–2 ms–1
Answer
C
Question. An electron of mass m, when accelerated through a potential difference V, has de Broglie wavelength λ. The de Broglie wavelength associated with a proton of mass M accelerated through the same potential difference, will be
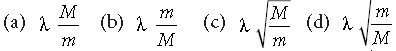
Answer
D
Question. If particles are moving with same velocity, then which has maximum de Broglie wavelength?
(a) proton
(b) a-particle
(c) neutron
(d) b-particle
Answer
D
Question. An electron of mass m with an initial velocity v→ = v0^i (v0 > 0 ) enters an electric field E→ = −E→0 i^ (E0 = constant > 0) at t = 0. If λ0 is its de-Broglie wavelength initially, then its de- Broglie wavelength at time t is
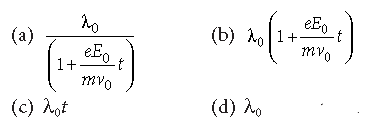
Answer
A
Question. The wavelength λe of an electron and λp of a photon of same energy E are related by

Answer
C
Question. The de-Broglie wavelength of neutrons in thermal equilibrium at temperature T is
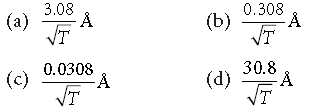
Answer
D
Question. An electron beam has a kinetic energy equal to 100 eV. Find its wavelength associated with a beam, if mass of electron = 9.1 × 10–31 kg and 1 eV = 1.6 × 10–19 J. (Planck’s constant = 6.6 × 10–34 Js)
(a) 24.6 Å
(b) 0.12 Å
(c) 1.2 Å
(d) 6.3 Å
Answer
C
Question. An a-particle moves in a circular path of radius 0.83 cm in the presence of a magnetic field of 0.25 Wb/m2. The de Broglie wavelength associated with the particle will be
(a) 1 Å
(b) 0.1 Å
(c) 10 Å
(d) 0.01 Å
Answer
D
Question. If the momentum of an electron is changed by P, then the de Broglie wavelength associated with it changes by 0.5%. The initial momentum of electron will be
(a) 200P
(b) 400P
(c) P/200
(d) 100P
Answer
A
Question. In the Davisson and Germer experiment, the velocity of electrons emitted from the electron gun can be increased by
(a) increasing the potential difference between the anode and filament
(b) increasing the filament current
(c) decreasing the filament current
(d) decreasing the potential difference between the anode and filament.
Answer
A
Assertion-Reason Questions
In the following questions, a statement of Assertion (A) is followed by a statement of Reason (R). Choose the correct answer out of the following choices.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false and R is also false.
Question. Assertion (A): An electron and a photon possessing same wavelength, will have the same momentum.
Reason (R): Momentum of both particle is same by de Broglie hypothesis.
Answer
A
Question. Assertion (A): Matter has wave-particle nature.
Reason (R): Light has dual nature.
Answer
B
Question. Assertion (A): Photoelectric process is instantaneous process.
Reason (R): When photons of energy (hn) greater than work function of metal (φ0) are incident on a metal, the electrons from metal are emitted with no time lag.
Answer
A
Question. Assertion (A): Photoelectric effect demonstrates the wave nature of light.
Reason (R): The number of photoelectrons is proportional to the velocity of incident light.
Answer
D
Question. Assertion (A): Threshold frequency depends on intensity of light.
Reason (R): Greater is the photon frequency, smaller is the energy of a photon.
Answer
D
Question. Assertion (A): On increasing the intensity of light the photocurrent increases.
Reason (R): The photocurrent increases with increase of frequency of light.
Answer
C
Question. Assertion (A): In the process of photoelectric emission, all emitted electrons have the same kinetic energy.
Reason (R): According to Einstein’s equation Ek = hν + φ0.
Answer
D
Question. Assertion (A): If intensity of incident light is doubled, the kinetic energy of photoelectron is also doubled.
Reason (R): The kinetic energy of photoelectron is directly proportional to intensity of incident light.
Answer
D
Question. Assertion (A): The electrons and protons having same momentum has same de Broglie wavelength.
Reason (R): de Broglie wavelength λ = h/p
Answer
A
Question. Assertion (A): On increasing the frequency of light, the photocurrent remains unchanged.
Reason (R): Photocurrent is independent of frequency but depends only on intensity of incident light.
Answer
A

We hope you liked the above provided MCQ Questions Chapter 11 Dual Nature of Radiation and Matter Class 12 Physics with solutions. If you have any questions please ask us in the comments box below.


