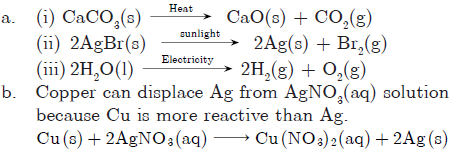Please refer to the Chemical Reactions And Equations Revision Notes given below. These revision notes have been designed as per the latest NCERT, CBSE and KVS books issued for the current academic year. Students will be able to understand the entire chapter in your class 10th Science book. We have provided chapter wise Notes for Class 10 Science as per the latest examination pattern.
Revision Notes Chapter 1 Chemical Reactions And Equations
Students of Class 10 Science will be able to revise the entire chapter and also learn all important concepts based on the topic wise notes given below. Our best teachers for Grade 10 have prepared these to help you get better marks in upcoming examinations. These revision notes cover all important topics given in this chapter.
ONE MARK QUESTIONS
Question: In electrolysis of water, why is the volume of gas collected over one electrode double that of gas collected over the other electrode.
Answer: Water contains hydrogen and oxygen in the ratio of 2 : 1, that is why volume of H2 is double than that of oxygen.
Question: Which one is a chemical change: Rusting of iron or melting of iron?
Answer: Rusting of iron is a chemical change.
Question: What can be seen when a strip of copper metal is placed in a solution of silver nitrate?
Answer: The solution will become blue, shiny silver metal will get deposited.
Cu(s) + 2AgNO3(aq) → Cu(NO3)2 (aq) + 2Ag(s)
Question: State one basic difference between a physical change and a chemical change.
Answer: In a physical change, no new substance is formed.
In a chemical change, new substance(s) with new properties is/are formed.
Question: State one industrial application of reduction process.
Answer: It is used in the extraction of metals e.g.,
ZnO(s) + C(s) Heat→ Zn(s) + CO2(g)
Question: Which one of the following is a chemical change? Give reason also.
(a) Burning of wax (b) Melting of wax
Answer: Burning of gas is a chemical change because new products with new properties will be formed on burning.
Question: Name the oxidising and reducing agent in the following reaction:
CuO + H2 → Cu + H2O
Answer: CuO is oxidising agent H2 is reducing agent.
Question: Write a complete balanced chemical equation for the following reaction:
Sodium hydroxide + Sulphuric acid →
Answer: 2NaOH + H2SO4 → Na2SO4 + 2H2O
Question: What is meant by a chemical reaction?
Answer: Chemical reaction, a process in which one or more substances, the reactants, are converted to one or more different substances, the products. Substances are either chemical elements or compounds. A chemical reaction rearranges the constituent atoms of the reactants to create different substances as products
Question: AgNO3(aq) + NaCl(aq) → AgCl ↓ + NaNO3(aq)
FeS + H2SO4 → FeSO4 + H2S ↑
Consider the above mentioned two chemical equations with two different kinds of arrows (- and . ) along with the product. What do these two different arrows indicate?
Answer: ↑ shows evolution of gas, ↓ represents a precipitate.
Question: Name and state the law which is kept in mind when we balance chemical equations.
Answer: Law of conservation of mass. It states Matter can neither be created nor be destroyed. The total mass of reactants must be equal to total mass of products.
Question: If copper metal is heated over a flame it develops a coating. What is the colour and composition of coating?
Answer: Black coloured coating is formed. It is a due to formation of copper oxide.
Question: Hydrogen being a highly inflammable gas and oxygen being a supporter of combustion, yet water, a compound made up of hydrogen and oxygen is used to extinguish fire. Why?
Answer: H2O is a compound constituted of hydrogen of oxygen elements and being a compound it has different properties as compared to its constituting elements.
Question: Write a balanced chemical equation to represent the following reaction: carbon monoxide reacts with hydrogen gas at 340 atm to form methyl alcohol.
Answer: CO(g) + 2H2(g) 340 atm → CH3OH(l)
Question: Is burning of a candle wax a physical or a chemical change?
Answer: Burning of a candle wax is a chemical change.
TWO MARKS QUESTIONS
Question: Balance the following chemical equation:
Fe(s) + H2O(g) → Fe3O4(s) + H2(g)
Answer: 3Fe(s) + 4H2O(g) → Fe3O4(s) + 4H2(g)
Question: Why is respiration considered as exothermic process?
Answer: Download more materials in free at : Respiration is an exothermic process because energy
is given out in respiration.
Question: Translate the following statements into chemical equations and then balance them:
a. Hydrogen gas combines with nitrogen to form ammonia.
b. Hydrogen sulphide gas burns in air to give water and sulphur dioxide.
c. Barium chloride reacts with aluminium sulphate to give aluminium chloride and a precipitate of barium sulphate. State the two types in which this reaction can be classified.
d. Potassium reacts with water to give potassium hydroxide and hydrogen gas.
Answer:
a. 3H2(g) + N2(g) → 2NH3(g)
b. 2H2S(g) + 3O2(g) → 2H2O(l) + 2SO2(g)
c. 3BaCl2(aq) Al2(SO4)3(aq) 3BaSO4(s) 2AlCl3(aq)
This reaction can be classified as a double displacement reaction as the two reacting species (BaSO4, Al2SO4)3 undergoes mutual exchange
of ions. It can also be classified as precipitation reaction, since a white ppt. of BaSO4 is obtained.
d. 2K(s) + 2H2O(l) → 2KOH(aq) + H2(g)
Question: Write balanced equation for the reaction between Mg and hydrochloric acid. Name the product obtained, identify the type of reaction.
Answer: Mg(s) + 2HCl(dil) → MgCl2(aq) + H2(g)
Magnesium chloride and hydrogen gas are formed in this reaction. It is a displacement reaction.
Question: Why are decomposition reactions called the opposite of combination reactions? Give chemical equations for these reactions.
Answer: In decomposition reactions, a compound is broken down into one or more elements or compounds e.g.,
CaCO3(s)→ Heat CaO(s) + CO2(g)
In combination reactions, two or more elements or compounds combine to form a new compound.
Therefore, decomposition reactions are opposite to combination reactions e.g.,
3Mg(s) + N2(g) Burns Mg3N2(s)
Question: When a metal X is added to salt solution of metal Y, the following chemical reaction takes place:
Metal X + Salt solution of Y → Salt solution of X + Metal Y
Mention the inference you draw regarding the reactivity of metal X and Y and also the type of reaction. State the reason of your conclusions.
Answer: X is more reactive than Y because , X displaces Y from its salt solution. Hence, this reaction is an example of displacement reaction.
63. Why does the colour of copper sulphate solution changes when an iron pin is dipped in it?
Answer: Iron being more reactive displaces copper from copper sulphate (Blue) solution to form iron (II) sulphate
(Pale green) solution and reddish brown copper metal gets deposited.
Fe(s) + CuSO4 → FeSO4(aq) + Cu(s)
Question: A solution of potassium chloride when mixed with silver nitrate solution, an insoluble white substance is formed. Write the chemical reaction involved and also mention the type of reaction.
Answer:
AgNO3(aq) + KCl(aq) → AgCl(s)+KNO3(aq)
(White ppt.)
It is a double displacement reaction.
Question: Using a suitable chemical equation justify that some chemical reactions are determined by
a. Change in colour.
b. Change in temperature.
Answer: a. Change in colour: Blue coloured crystalline copper sulphate crystals changes to white CuSO4
(anhydrous) on heating.
CuSO4.5H2O → Heat CuSO4 + 5H2O
(Blue) (Dirty white)
b. Change in temperature:
CaO(s) + H2O(l) → Ca(OH)2(aq) + Heat
Question: Identify the type of each of the following reactions:
a. A reaction in which a single product is formed from two or more reactants.
b. The reaction mixture becomes warm.
c. An insoluble substance is formed.
d. External surface of the container in which reaction takes place becomes cold.
Answer: a. Combination reaction
b. Exothermic reaction
c. Precipitation reaction (Double displacement reaction)
d. Endothermic reaction
Question: Balance the following reactions:
a. BaCl2 + H2SO4 → BaSO4 + HCl
b. Ca(OH)2 + HNO3 → Ca(NO3)2 + H2O
c. Pb(NO3)2 → PbO + NO2 + O2
d. MnO2 + HCl → MnCl2 + Cl2 + H2O
Answer:
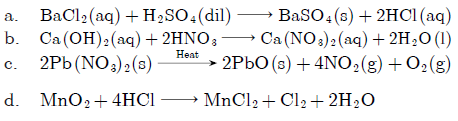
Question: Write the balanced equation for the following reactions and identify the type of reaction in each case:
a. Potassium bromide + Barium iodide →
Barium bromide + Potassium Iodide
b. Hydrogen(g) + Chlorine(g) → Hydrogen
chloride(g)
Answer:

Question: (a) A solution of substance X is used for white washing. What is substance X? Write the chemical reaction of X with water.
(b) Why does the colour of copper sulphate solution changes when iron nail is dipped in it?
Answer: a. X is CaO, calcium oxide
CaO(s) + H2O(l) → Ca(OH)2(aq)
b. It is because Fe displaces Cu from CuSO4 (blue)
solution to form FeSO4 (pale green) and reddish brown Cu metal gets deposited.
Question: A white salt on heating decomposes to give brown fumes and a white residue is left behind.
a. Name the salt.
b. Write the equation for the decomposition reaction.
Answer:
a. Lead nitrate
b. 2Pb(NO3) 3(s)→ Heat 2PbO(s) 4NO2 (g)+ O2 (g)
(White) Residue Brown
Question: When a solution of potassium iodide is added to a solution of lead nitrate in a test tube, a reaction takes place.
a. What type of reaction is this?
b. Write the balanced chemical equation to represent the above reaction.
Answer: a. It is precipitation as well as double displacement reaction.
b. Pb(NO3) (aq)+2KI (aq) Pbl2→(s)+2KNO3(aq)
Question: A zinc plate was put into solution of copper sulphate kept in a glass container. It was found that blue colour of the solution gets fader and fader with passage of time. After few days when zinc plate was taken out of the solution, a number of holes were observed on it. a. State the reason for the changes observed on zinc plate.
b. Write the chemical equation for the reaction involved.
Answer: a. Zinc displaces copper from copper sulphate
solution to form colourless ZnSO4 and copper metal is deposited. Zinc gets consumed due to which holes are formed.
b. Zn(s) + CuSO4(aq) → ZnSO4(aq) + Cu(s)
Question: Define combination reaction. Give one example of a combination reaction which is also exothermic.
Answer: Combination reaction: The reaction in which two or more elements or compounds combine to form compound(s) e.g.,
C(s) + O2(g) → CO2(g) + Heat
It is an exothermic, combination reaction.
Question: What happens when an aqueous solution of sodium sulphate reacts with an aqueous solution of barium chloride? State the physical conditions of reactants in which the reaction between them will not take place. Write the balanced chemical equation for the reaction and name the type of reaction.
Answer: White precipitate of BaSO4 is formed.
BaCl2(aq)+ Na2SO4(aq) → BaSO4(s)+ 2NaCl (aq)
It is a double displacement reaction. If reactants are taken in solid state, products will not be formed.
Question: What is redox reaction? When a magnesium ribbon burns in air with a dazzling flame and forms a white ash, is magnesium oxidised or reduced. Why?
Answer:Redox reaction is a reaction in which oxidation and reduction takes place simultaneously.
Mg is getting oxidised because it is gaining oxygen to form magnesium oxide.
Question: Write any two observations in an activity which may suggest that a chemical reaction has taken place. Give an example in support of your answer.
Answer:

Question: When the powder of common metal is heated in open china dish its colour turns black. However, when hydrogen is passed over the hot black substance so formed, it regains its original colour. Based on the Download more materials in free at : above information answer the following questions:
a. What type of chemical reaction takes place in each of the two given steps?
b. Name the metal initially taken in powdered form. Write balanced equations for both the reactions.
Answer

Question: Why do we store silver chloride in dark coloured bottle? Explain in brief.
Answer: It is done so as to cut off the exposure to sunlight.
AgCl is photosensitive, it will decompose to Ag and Cl2 in the presence of sunlight.
2AgCl(s) → sunlight 2Ag(s) + Cl2( g)
Question: “Oxidation and reduction processes occur simultaneously”. Justify this statement with the help of example.
Answer: Oxidation involves loss of electrons or addition of oxygen. Whereas reduction involves gam of electrons
or addition of hydrogen e.g.,
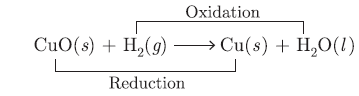
H2 is getting oxidised to H2O, CuO is getting reduced to Cu.
It shows oxidation and reduction occur simultaneously.
Question: When magnesium ribbon bums in air or oxygen, a product is formed. State the type of chemical reaction and name the product formed in the reaction. Write balanced chemical equation for the reaction.
Answer:It is a combination reaction. The product formed is magnesium oxide:
2Mg(s) + O2(g) → 2MgO(s)
Question: Distinguish between a displacement reaction and a double displacement reaction. Identify the displacement and the double displacement reaction from the following reactions:
a. HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(l)
b. Fe(s) + CuSO4(aq) → FeSO4(aq) + Cu(s)
Answer: Displacement Reaction: When a more reactive metal displaces a less reactive metal from its salt solution.
Double displacement reaction: When two compounds exchange their ions to form two new compounds.
a. Double displacement reaction.
b. Displacement reaction.
Question: What is an oxidation reaction? Give an example of oxidation reaction. Is oxidation an exothermic or an endothermic reaction?
Answer:
Oxidation reaction: The reaction in which O2 is added
or H2 is removed or loss of electrons takes place is
called oxidation reaction. E.g.,
2Cu(s) + O2(g) → 2CuO(s)
It is an exothermic reaction.
THREE MARKS QUESTIONS
Question: Define the term decomposition reaction. Give one example each of thermal decomposition and electrolytic decomposition reactions.
Answer: Decomposition reaction: The reaction in which a compound is broken down into simpler elements or compounds:
a. CaCO3(s) → Heat CaO(s) + CO2(g)
b. 2H2O(l)→ Electricity 2H2(g) + O2(g)
Question: Name two salts that are used in black and white photography. Give equations for the reactions when these are exposed to sunlight.
Answer: AgBr (Silver bromide) and AgI (Silver iodide) are
used in black and white photography.
2AgBr(s)→ sunlight 2Ag(s) + Br2(g)
2AgI(s) → sunlight 2Ag(s) + I2(g)
Question: Write the chemical equation of the reaction in which the following changes take place with an example of each.
a. Change in colour,
b. Change in tem-perature,
c. Formation of precipitate.
Answer:

Question: State the type of chemical reactions and chemical equations that take place in the following:
a. Magnesium wire is burnt in air.
b. Electric current is passed through water.
c. Ammonia and hydrogen chloride gases are mixed.
Answer:
a. 2Mg(s) + O2(g) → 2MgO(s);
b. Oxidation reaction
c. 2H2O(l) → 2H2(g) + O2(g);
Decomposition reaction
d. NH3(g) + HCl(g) → NH4Cl(s);
Combination reaction
Question: Write the balanced chemical equations for the following chemical reactions:
a. Hydrogen + Chlorine → Hydrogen Chloride
b. Lead + Copper Chloride → Lead chloride + Copper
c. Zinc oxide + Carbon → Zinc + Carbon Monoxide
Answer:
a. H2(g) + Cl2(g) → 2HCl(g)
b. Pb(s) + CuCl2(aq) → PbCl2(s) + Cu(s)
c. ZnO(s) + C(s) → Zn(s) + CO(g)
Question: Write chemical equations for the reactions taking place when a. Iron reacts with steam
b. Magnesium reacts with dil. HCl
c. Copper is heated in air
Answer:
a. 3Fe(s) + 4H2O(g) → Fe3O4(s) + 4H2(g)
b. Mg(s) + 2HCl(dil) → MgCl2(aq)
c. 2Cu(s) + O2(g) → 2CuO(s)
Question:(i) Solid calcium oxide was taken in a container and water was added slowly to it.
(a) Write the observations.
(b) Write the chemical formula of the product formed.
(ii) What happens when carbon dioxide is bubbled through lime water (a) in small amount (b) in excess?
Answer:
(i) (a) The container becomes hot and hissing sound is produced.
(b) Ca(OH)2 is the formula of the product formed.
(ii) (a) Lime water turn milky when CO2(g) is passed through it:
Ca(OH)2(aq) + CO2(g) → CaCO3(s) + H2O(l)
(b) If excess of CO2 is passed milkiness disappears:
CaCO3(s) + CO2(g) + H2O(s) → Ca(HCO3)2(aq)
Question: Write the balanced chemical equation for the following
Download more materials in free at : reaction and identify the type of reaction.
Thermite reaction, iron (III) oxide reacts with aluminium and give molten iron and aluminium oxide.
Answer:
Fe2O3(s) + 2Al(s) → Al2O3(s) + 2Fe(l)
It is displacement as well as redox reaction.
Question: An aqueous solution of metal nitrate ‘P’ reacts with sodium bromide solution to form yellow precipitate
‘Q’ which is used in photography. ‘Q’ on exposure to sunlight undergoes decomposition to form metal present along with a reddish brown gas. Identify ‘P’ and ‘Q’ write the balanced chemical equation for the chemical reaction. List the two categories in which
reaction can be placed.
Answer:
‘P’ is silver nitrate (AgNO3)
AgNO3(aq) + NaBr(ag) → AgBr(s) + NaNO3(aq)
(P) (Yellow ppt)
This reaction is categorised as double displacement as well as precipitation reaction.
2AgBr(s)→ sunlight 2Ag(s) + Br2(g)
The above reaction is photochemical decomposition reaction.
FIVE MARKS QUESTIONS
Question: Identify the type of reactions taking place in each of the following:
a. Barium chloride solution is mixed with copper sulphate solution and white precipitate is formed.
b. On heating copper powder in china dish, the surface of copper powder turns black.
c. On heating green coloured ferrous sulphate crystals, raddish brown solid is left and smell of a gas having odour of burning sulphur is experienced.
d. Iron nails when left dipped in blue copper sulphate solution become reddish brown in colour and the blue colour of copper sulphate fades away.
e. Quick lime reacts vigorously with water releasing
a large amount of heat.
Answer:
a. Double displacement reaction,
b. Oxidation,
c. Decomposition reaction,
d. Displacement reaction,
e. Combination reaction.
Question: a. Define a balanced chemical equation. Why should an equation be balanced?
b. Write a balanced chemical equation for the following reactions:
(i) Phosphorus burns in the presence of chlorine
to form phosphorus pentachloride.
(ii) Burning of natural gas.
(iii) The process of respiration.
Answer:
a. Balanced chemical equation is a equation in which number of atoms of various elements are equal on both sides of the equation.
The equation should be balanced due to law of conservation of mass.
b. (i) 2P(s) + 5Cl2(g) → 2PCl5(S)
(ii) CH4(g) + O2(g) → CO2(g) + 2H2O(l)
(iii) C6H12O6(s) + 6O2(g) → 6CO2(g) + 6H2O(l)
Question: (a) Write one example for each of decomposition reaction carried out with the help of (i) Electricity,
(ii) Heat, (iii) Light.
(b) Which of the following statement is correct and why?
Copper can displace silver from silver nitrate solution and silver can displace copper from copper sulphate solution.
Answer: