Please see Carbon and Its Compound Class 10 Science Revision Notes provided below. These revision notes have been prepared as per the latest syllabus and books for Class 10 Science issues by CBSE, NCERT, and KVS. Students should revise these notes for Chapter 4 Carbon and Its Compound daily and also prior to examinations for understanding all topics and to get better marks in exams. We have provided Class 10 Science Notes for all chapters on our website.
Chapter 4 Carbon and Its Compound Class 10 Science Revision Notes
Why Carbon forms Covalent Bonds
We know that a majority of substances used by us daily, from paper and plastics to coal and petrol, are all made up of carbon. Food grains, pulses, medicines, cotton, synthetic fibres, wood etc. are all made up of carbon. Carbon is also a major part of all living things. In air, it is present as carbon dioxide and comprises around 0.03% of the total air.
Let us study about carbon and its bonding in its compound in more detail.
Carbon is a non-metal having the symbol ‘C’ and atomic number six. Since the atomic number of carbon is six, its electronic configuration is 2, 4. This means that carbon contains two electrons in K shell and 4 electrons in L shell (outermost shell). Hence, it has four electrons in its valence shell.
Since carbon has four electrons in its valence shell, it needs four more electrons to complete its octet. Therefore, it is a tetravalent element.
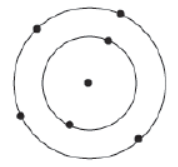
In order to complete its octet i.e., to attain its noble gas configuration and to stabilize itself, carbon can:
- Either lose four electrons to form C4+ or gain four electrons to form C4−. This, however, requires a lot of energy and would make the system unstable.
Therefore, carbon completes its octet by sharing its four electrons with the other carbon atoms or with atoms of other elements.
The bonds that are formed by sharing electrons are known as covalent bonds. Covalently bonded molecules have weak intermolecular forces, but intramolecular forces are strong.
Carbon has four valence electrons and needs four more electrons to complete its octet. Therefore, it is capable of bonding with four other atoms of carbon or atoms of other elements having a valency of 1.
For example, the molecule, methane can be formed with hydrogen (H) atoms that have only one electron in its K shell. To attain the noble gas configuration, it combines with four hydrogen atoms as shown in the figure.
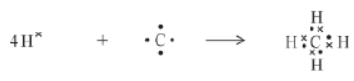
Now, let us study the properties of carbon compounds.
- Covalent bonds are made by atoms by sharing their electrons. Formation of ions does not take place in this process. In addition, these compounds do not have any extra electrons.
Hence, covalent compounds are non- conductors of electricity.
- As all organic compounds contain covalent bonds, they also have low melting and boiling points. This becomes evident from the following data.
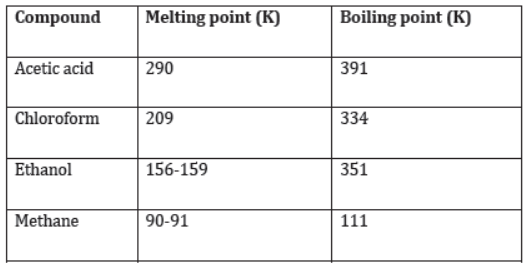
Also, from the above data, it can be inferred that the forces of attraction between the carbon molecules in carbon compounds is not very strong.
Because of their low melting and boiling points, these compounds mostly exist as liquids or gases at room temperature.
Allotropic Forms of Carbon
Carbon is the versatile element present in food, clothes, medicines, papers, etc. In addition, all living structures are carbon based. The earth’s crust has only 0.02% carbon in the form of minerals and the atmosphere has 0.03% carbon dioxide. In spite of the small amount of carbon available in nature, the importance of carbon is immense.
Position of carbon in the periodic table
Carbon has an atomic number 6 and an atomic mass as 12 u. It contains 2 and 4 electrons in the K and L shell respectively. It belongs to the group IVA of the periodic table and has a valency of 4.
Occurrence: Carbon occurs in free as well as in combined state.
In free state, carbon occurs as diamond, graphite, and coal. Diamond and graphite are pure forms of carbon while coal is an impure form of carbon in which the percentage of carbon varies from 24% to 90%.
In combined state, carbon occurs in
- bio-molecules such as cellulose, carbohydrates, fats, proteins, etc.
- minerals in the form of carbonates
- calcium carbonate (CaCO3) in lime, marble, and chalk
- magnesium carbonate (MgCO3) in magnesite
- calcium and magnesium carbonate (MgCO3.CaCO3) in dolomite
- calamine (ZnCO3) as zinc carbonate Also,
- It is present in large amount in petrol, kerosene oil, diesel oil, grease, and wax.
- Natural gas, marsh gas, petroleum gas, and coal gas contain carbon in the form of its compounds.
Allotropy: The phenomenon due to which an element exhibits different physical forms is called allotropy.
Reasons for allotropy are different arrangement of atoms in each allotrope, different methods by which each allotrope is prepared, and different amount of energy associated in
the formation of each allotrope.
Let us study about the different allotropes of carbon.
Carbon is considered to have two main kinds of allotropes i.e., crystalline and amorphous.
Following is a classification of the crystalline and amorphous forms of carbon:
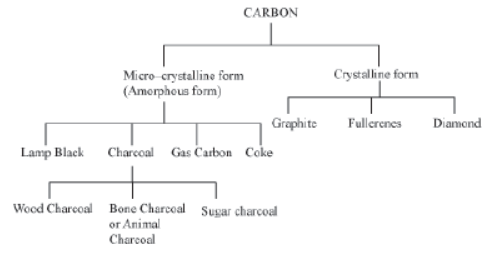
Well known allotropes of carbon are:
Among these, diamond and graphite have been known for a very long time and are quite common. Buckminsterfullerene, on the other hand, was discovered just recently. Thus, not much is known about it.
Crystalline forms of carbon
Diamond
Crystal structure: In diamond, each atom of carbon of the given crystal unit is surrounded by four other carbon atoms, which are joined by covalent bonds, such that they form a regular tetrahedron. The diamond crystal is a compact structure in which atoms of a single unit lie in different planes. Thus, the atoms cannot slip because of their different positions in different planes and hence, diamond is the hardest naturally occurring substance.
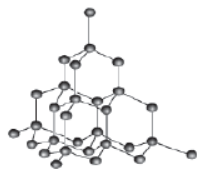
Physical properties: The physical properties of diamond are:
- It is the hardest naturally occurring substance.
- Pure diamond is colourless, transparent, but brittle solid.
- It is chemically inert under ordinary conditions as there are no free electrons available.
- It is a good conductor of heat but is does not conduct electricity.
- Its refractive index is 2.42.
- Its specific gravity is 3.52.
- It is the densest form of carbon, its density being 3.5.
- It is transparent to X-rays, ultraviolet rays, and visible light rays.
Chemical properties: The chemical properties of diamond are:
- Action of air or oxygen: When diamond is heated in air, it catches fire at 800oC. It does not leave behind any ash and therefore, it is the purest form of carbon.

- Action of sodium carbonate: When diamond is heated very strongly with solid sodium carbonate, it reacts to form sodium oxide and carbon monoxide.

Uses: The various uses of diamonds are:
- They are generally used for making ornaments.
- They are used for cutting glass as well as diamonds.
- They are used for making dies for drawing wires.
- They are used as bearings in watches.
- They are used in making laser beam in electronics.
- Diamond is a poor conductor of electricity.
Can you tell why?
All 4 valence electrons in diamond are involved in bonding. Therefore, these are immobile and do not conduct.
Graphite
Crystal structure: In graphite, atoms of a single crystal are arranged in a hexagonal ring in a single plane. The bonds between the carbon atoms of two single crystals in the parallel planes are weak. Thus, one plane can easily slide over another plane by applying pressure.
This is why graphite is soft and can be used as a lubricant.

Physical properties: The physical properties of graphite are:
- It is dark grey solid, having lustre.
- It is a very good conductor of heat and electricity.
- It is opaque to light.
- Its specific gravity is 2.2.
- It is opaque to X-rays, ultraviolet rays, and visible light rays.
Chemical properties: The chemical properties of graphite are:
- Action of air or oxygen: Graphite does not burn in air. However, if graphite is heated in air, then it catches fire at 700oC and forms a mixture of carbon dioxide and carbon monoxide.
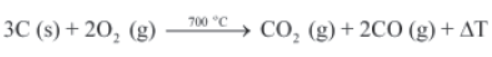
- Action of sodium carbonate: Like diamond, graphite also form sodium oxide and carbon monoxide when heated with solid sodium carbonate.

Uses: The various uses of graphite are:
- It is used in making lead pencils.
- It is used in making electrodes.
- It is used as a dry lubricant.
- It is used as a moderator in a nuclear reactor.
Buckminsterfullerene:
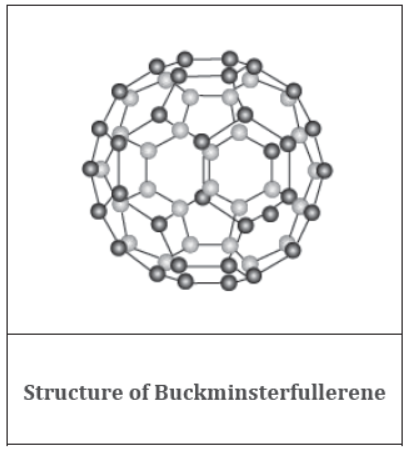
It is the third and the most recently discovered allotrope of carbon.
Buckminsterfullerene is a cluster of sixty carbon atoms arranged in the form of a football.
It is named after the American architect Buckminster Fuller, as it resembled the geodesic dome designed by him. Since it contains sixty carbon atoms, its chemical formula is C60.
Amorphous Forms of Carbon
What is an amorphous solid?
An amorphous solid is a non-crystalline solid with no well-defined ordered structure.
Some amorphous forms of carbon:
- Charcoal: It is produced from the bones of animals, combustion of wood etc.
- Lampblack or soot: It is produced from the incomplete combustion of hydrocarbons.
- Coal: It is formed by decomposition of dead remains of plants when they got buried in the land owing to the high temperature and pressure conditions present there. Based on the content of carbon present, normally three types of coal deposits exist and these are as follows.
- Lignite with 25-35% of carbon content.
- Anthracite with carbon content of more than 80%.
- Bituminous coal with 60-80% carbon
- Peat with less than 60% carbon content.
- Uses of Coal:
- As a fuel for cooking, in industries
- For heating of bricks
- In thermal power plants to generate electricity
- Coke: It is a tough, porous, black substance. It is an almost pure form of carbon obtained from coal.
- Ues of coke:
- In the manufacture of steel
- In the extraction of many metals.
Bonding In Carbon Compounds
Do you know that the number of carbon compounds outnumber the compounds of all the other elements taken together? The number of carbon compounds is so vast that there is one branch of chemistry dedicated only to the study of carbon and its compounds. This branch is known as organic chemistry.
Do you know why carbon is a part of so many compounds? Which properties of carbon help it to be a part of so many compounds?
Here, we will try to find the answers to these questions.
The most important property of carbon that allows it to be a part of so many compounds is catenation. Catenation is the ability of an element to combine with itself through covalent bonds.
Carbon has a unique ability to combine with other atoms of carbon to form long chains and rings. Carbon forms very strong bonds with itself. Hence, the chains and rings formed by carbon through catenation are very stable. Thus, these chains and rings are not only long, but also very stable. This is because carbon can form strong single, double, and triple bonds with other atoms of carbon. This can give rise to branches, chains, and rings as shown in the figure below.

Formation of long chained, branched, and cyclic structures of carbon.
Now, carbon has a valency of four. This means that it can bond with four other carbon atoms or four other monovalent atoms, which gives rise to compounds having specific properties. These properties depend on carbon as well as on elements other than carbon.
Do You Know:
Apart from carbon, a very few elements display the property of catenation so extensively.
Silicon can join with other atoms of silicon, but the maximum length of the chains that can be obtained in the case of silicon is limited to seven or eight atoms. In addition, the
compounds formed by silicon by catenation are not very stable.
Saturated and Unsaturated Compounds
Saturated compounds:
- The compounds of carbon having only single bonds between the carbon atoms are called saturated compounds.
For example, a molecule of ethane (C2H6) contains one single bond between the carbon atoms.


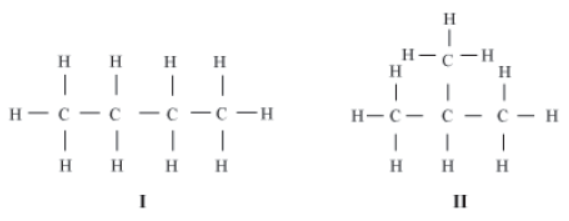
Similarly, butane (C4H10) can exist in two different structures, I and II. Both these structures contain three single bonds between the carbon atoms.
Unsaturated compounds:
- The compounds of carbon having double and triple bonds between the carbon atoms are called unsaturated compounds.
For example, ethene is an unsaturated compound containing one double bond between the carbon atoms.

Similarly, ethyne is an unsaturated compound containing one triple bond between the carbon atoms.


The chains, branches, and rings that carbon forms by forming bonds with it can be saturated as well as unsaturated.
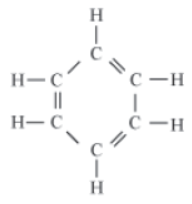
For example, benzene is a six-member ring that contains three double bonds. Thus, it is an unsaturated hydrocarbon
Organic compounds and Homologous Compounds
The number of carbon compounds is so vast that there is one branch of chemistry dedicated only to the study of carbon and its compounds. This branch is known as organic chemistry.
The compounds of carbon having only single bonds between the carbon atoms are called saturated compounds and those that contain a double or a triple bond between any two carbon atoms are known as unsaturated compounds.
For example, a molecule of propane (C3H8) contains two single bonds between the carbon atoms.
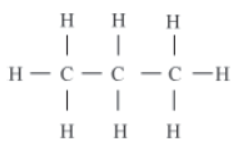
There are two main groups of hydrocarbons:
Aliphatic hydrocarbons:
- They are derived from fossil fuels.
- They are those hydrocarbons which possess straight chain or branched chain structures.
- They are grouped as:
- Alkanes: The general formula of alkanes is CnH(2n+2), n = number of carbon atoms.
- They contain only single bonds between all the carbon atoms present in a given molecule of an alkane.
- For example, methane (CH4), ethane (C2H6) etc.
- Alkenes: The general formula of alkenes is CnH2n, where n = number of carbon atoms
- They contain at least one carbon − carbon double bond.
- For example, ethene (C2H4), propene (C3H6) etc.
- Alkynes: The general formula of alkynes is CnH(2n−2), where n = number of carbon atoms
- They contain at least one carbon-carbon triple bond.
- For example, ethyne (C2H2), propyne (C3H4) etc.
Alicyclic saturated hydrocarbons:
- They are those saturated hydrocarbons in which the carbon atoms form a ring.
- Their general formula is similar to that of alkenes (CnH2n).
- For example, cyclopropane

Aromatic hydrocarbons :
Members of this group have a characteristic odour.
First recognised aromatic hydrocarbon is Benzene (C6H6)

- Benzene is a good organic solvent, which is used in the preparation of dyes, medicines, perfumes etc.
Consider, a molecule of butane (C4H10) contains three single bonds between the carbon atoms.
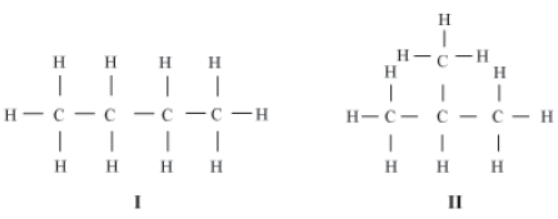
You must have noticed that these structures have carbon atoms linked to each other in a repeated manner. Such structures are called Chain structures and compounds exhibiting such structures are known as aliphatic compounds.
Do you observe anything in the structure of butane? The carbon atoms can be linked in two different ways (I and II). While I is a chain structure, II is a branched structure. Both have the same chemical formula, but they differ in their structures. Such compounds are called Structural isomers.
Carbon atoms in a molecule cannot only be linked in a chain or branch form, but these can also be arranged in a ring. For example, benzene, cyclohexane etc.
Benzene is a six-member ring that contains three double bonds. Thus, it is an unsaturated hydrocarbon. On the other hand, cyclohexane has only single bonds. Hence, it is a saturated compound.

However, the organic compounds which contain at least one benzene ring are known as aromatic compounds. For example, benzene, toluene, xylenes etc.
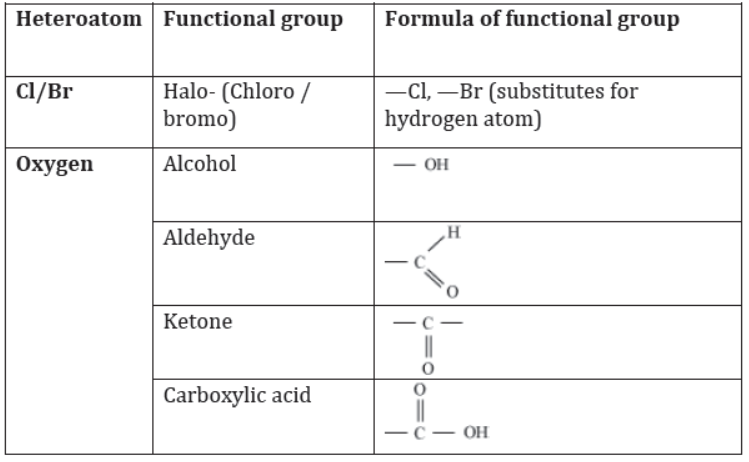
Carbon not only bonds to carbon and hydrogen, but it can also bond to other atoms like halogens (Br, Cl, I, F), nitrogen, oxygen etc. Sometimes, the hydrogen attached to a carbon in a hydrocarbon can also be replaced by another atom. In such cases, the replacing atom is called a heteroatom. A single atom or a group of atoms, which on bonding with a hydrocarbon, modifies the properties of the compound, is referred to as a functional group.
Thus, we see that carbon atoms can be linked to each other, or to hydrogen atoms, or to functional groups. In addition, the functional groups can change their properties.
However, it was noticed that the properties of CH3OH, C2H5OH, and C3H7OH are very similar. Can you tell why? This is because they form a part of the Homologous series.
Organic compounds that have similar general formula belong to a series called homologous series. The chemical properties of the compounds in the series are the same because of the presence of the same functional group. Therefore, the organic compounds containing same functional group are tested using same identification tests. On the other hand, the organic compounds with different functional groups will have different physical and chemical properties, hence different identification tests are needed to identify them.
Alkanes, alkenes, and alkynes form such series in which members differ in mass by 14 atomic mass units. For example, the alkane homologous series contains methane (CH4), ethane (C2H6), propane (C3H8), butane (C4H10), and pentane (C5H12) as its members. Each member differs from the previous one by a CH2 group (or 14 atomic mass units).
Let us see the first four members of the alkane family.
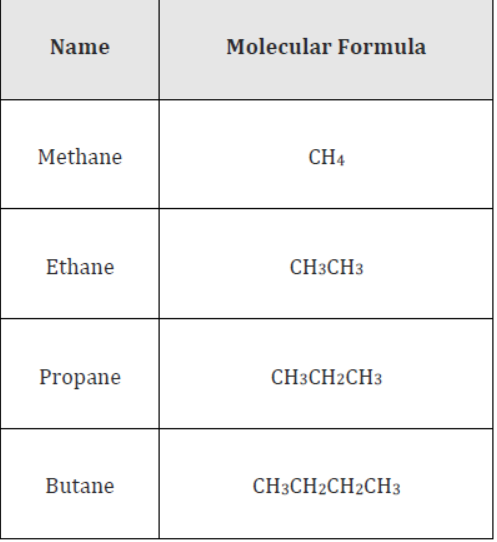
Thus, it is seen that any two consecutive members differ by a common difference of CH2 group. Hence, any two consecutive members differ in mass by 14 units.
Similarly, in alcohol homologous series, we have methanol (CH3OH), ethanol (C2H5OH), isopropanol (C3H7OH), and butanol (C4H9OH).
Organic compounds show an interesting phenomenon called STRUCTURAL ISOMERISM.
This phenomenon is shown by organic compounds with same molecular formula but different structural formula. These compounds are called ISOMERS. They have different properties from one another.
Structural isomerism arises due to the difference in the arrangement of atoms within the molecule. Types of isomerism:
a) Chain/ skeletal/ nuclear isomerism:
In this type, there is a difference in the structure of the carbon chain that forms the nucleus of the molecule. For example:

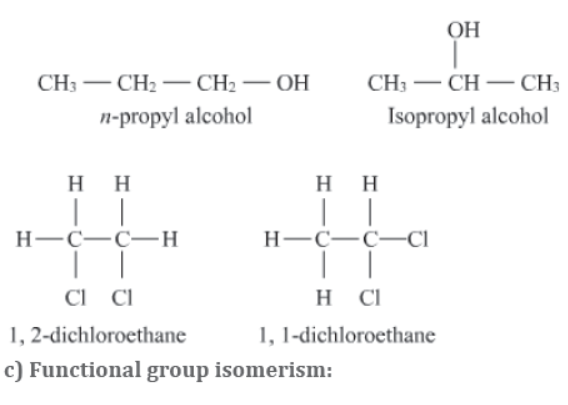
b) Position isomerism:
It arises due to the difference in the position of the functional group, the carbon-carbon multiple bonds or the substituent group. For example:
- It arises by virtue of the presence of different functional groups. For example:
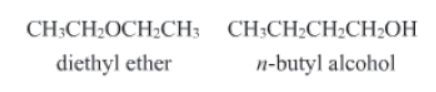
d) Metamerism:
In this type, there is a difference in the number of carbon atoms on either side of the functional group. For example:
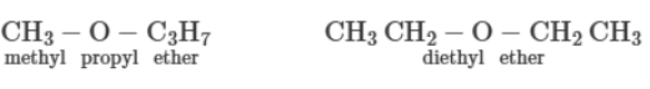
IUPAC Nomenclature of Carbon Compounds
Earlier, various organic compounds were known by their common names. However, the number of organic compounds increased at such a rapid pace that it became important to devise a method to name them scientifically to allow the scientists in every part of the world to understand their molecular formula. Hence, the International Union of Pure and Applied Chemistry (IUPAC) devised rules to name organic compounds. In this system of nomenclature, the names are correlated with the structures such that the learner can deduce the structure from the name. However, before the IUPAC system of nomenclature, organic compounds were assigned trivial or common names based on their origin or certain properties. For instance, citric acid is named so because it is found in citrus fruits. Also, the acid found in red ant is named formic acid since the Latin word for ant is ‘formica’.
Common or trivial names of some organic compounds

The naming of various compounds of carbon can be done as follows:
1. The longest continuous chain of carbon atoms should be identified first. This chain may or may not be a straight chain.
The base name of an organic compound depends upon the number of carbon atoms present in the chain. A compound having only one carbon atom starts with ‘meth-’, two carbon atoms start with ‘eth-’, and so on. The list of base names for one to ten carbon atoms is given in the following table.
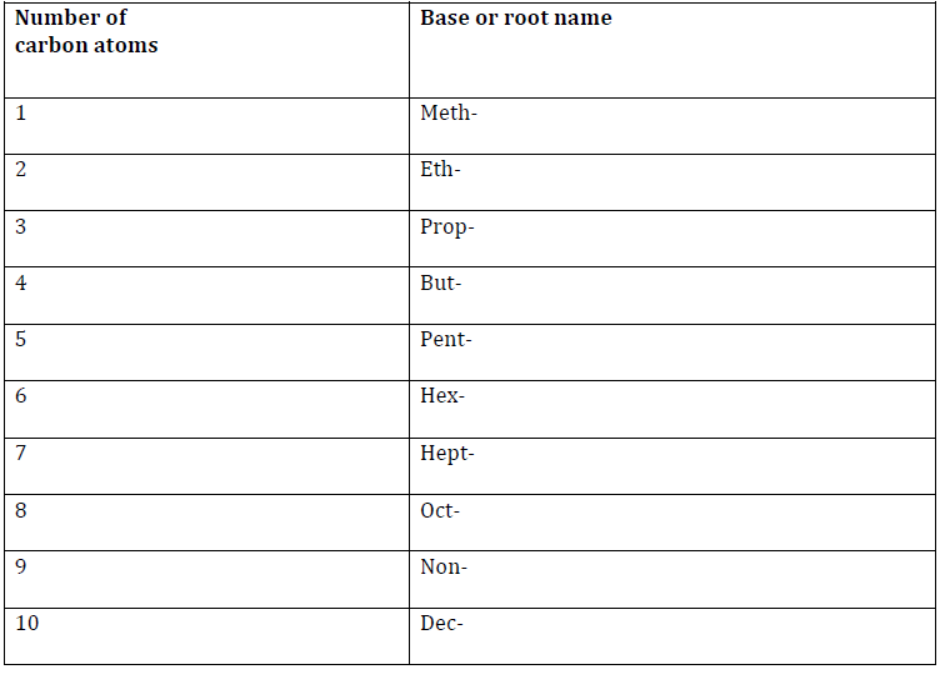
Base names according to the number of C atoms
2. The presence of a functional group is indicated by adding either a suffix or prefix to the base or root name.
If a hydrocarbon contains no double or triple bonds i.e., the hydrocarbon is saturated, then it is known as alkane and the suffix ‘-ane’ is added to the base word.
Therefore, CH4 becomes methane and CH3CH3 become ethane.
If a compound contains one or more double bonds, then it is known as alkene and the suffix ‘-ene’ is used.
If a compound contains one or more triple bonds, then it is called alkyne and the suffix ‘-
yne’ is used.
Similarly, the presence of other functional groups is indicated by adding different suffixes or prefixes to the root word. The following table shows the list of functional groups along with the suffixes/prefixes used to indicate their presence.
Prefixes/suffixes used to indicate different functional groups


If the last letter of a hydrocarbon is ‘e’ as in alkanes, then ‘e’ is deleted when the suffix for the functional group is added. For example, ‘e’ of pentane is deleted when the suffix ‘-one’ for the ketone group is added. Thus, we obtain pentanone from pentane and not pentaneone.
Hence, the name of each compound in a homologous series is based on the name of the basic carbon chain, modified by a prefix or a suffix, indicating the nature of the functional group.
Let us name a few compounds using IUPAC nomenclature.
Functional groups:
Functional groups are structural units within organic compounds that are defined by specific bonding arrangements between specific atoms.
Alcohol
If one hydrogen atom of an alkane is displaced by hydroxyl group(-OH), then alcohol compound is obtained. General formula of alcohol is CnH2n+1OH. Hence, it is represented as R-OH where R stands for alkyl group.
Aldehyde and Ketone
These are the functional groups containing carbonyl group. The carbon atom of carbonyl group is attached with one alkyl group and one hydrogen atom in case of aldehyde whereas, in case of a ketone carbon atom of carbonyl compound is attached with carbon atoms of two alkyl groups.
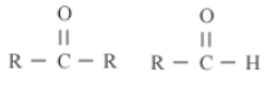
Carboxylic acid
Compounds having -COOH functional group are called carboxylic acid compounds. Some of them are ethanoic acid and propanoic acid. Carboxylic acid containing compounds have “oic” suffix. The general formula of carboxylic acid is
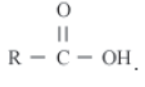
A systematic name of an organic compound is generally derived by identifying the parent hydrocarbon and the functional group(s) attached to it. See the example given below.
Let us try to name the following compounds.

Step − I: Select the longest carbon chain.
The longest possible chain of carbon atoms is picked and the compound is named as a derivative of this alkane.
Step − II: Assign the lowest number to the side chain.
The carbon chain selected is numbered from the end nearest to the side chain or the substituent so as to give the lowest number to the side chain.
Step-III: Arrangement of prefixes
In case more than one group is attached to the chain, these should be arranged alphabetically. Each substituent group is located on the parent chain by placing before it
the number of carbon atom to which it is attached.
Step-IV: Lowest number for functional group
In case some functional group is present in the chain, the lowest number is given to it. The lowest number is given in the following order of preference:
To the principle functional group of the compound
To the double or triple bond
To the substituent atoms or groups designated by prefixes
Therefore, the IUPAC name of the above organic compound is 3-Bromo-5-methylhexane.
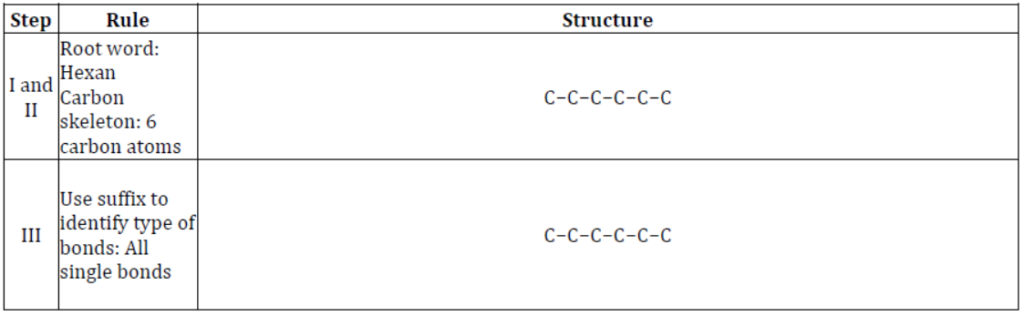
Conversely, using the IUPAC of an organic compound, it’s structure can be determined. The following rules help in accomplishing the task:
Step − I: Identify the root word. It forms the carbon skeleton in the structure.
Step − II: Write the number of carbon atoms as per the root word and number them from any end.
Step − III: As per the suffix in the name, ascertain the type of bond present in the compound. If any multiple bond is present, place it between the carbon atoms as stated in the IUPAC name.
Step − IV: Place the substituents at the carbon atoms mentioned in the IUPAC name.
Step − V: Place the functional group at the designated carbon atom.
Step − VI: Complete the valencies of the remaining carbon atoms by attaching hydrogen atoms.
For example, consider the given IUPAC name of an organic compound:
Hexan-3-ol
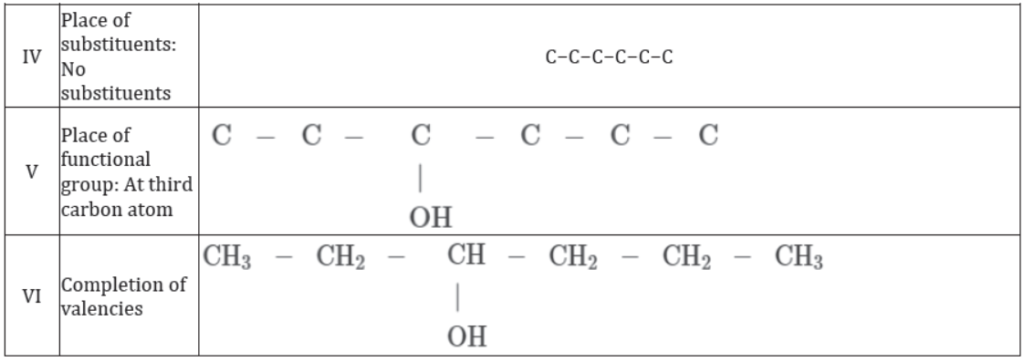
Combustion and Oxidation Reactions of Carbon Compounds and their Utility As Fuels
In our daily lives, we use a lot of organic compounds i.e., compounds that primarily contain carbon and hydrogen. Organic compounds such as coal and petroleum are of utmost importance in the modern world. These organic compounds are used as fuels to fulfil a majority of our energy requirements. Hence, it is important for us to know how these compounds react with other substances.
Do you know that organic substances burn to release heat and light energy?
We know that organic compounds primarily contain carbon and hydrogen. Therefore, hydrocarbons, when burnt, react with oxygen present in the air to give carbon dioxide and water vapours. A lot of heat and light energy is also released in the process. Such reactions are called combustion reactions.

Activity 1:
Take some carbon compounds such as naphthalene and camphor on a spatula and burn them one by one. Observe the nature of the flame produced by them when burnt.
It will be observed that on burning, camphor and naphthalene will give a yellow flame with lots of black smoke.
Carbon, in all its allotropic forms, burns in oxygen to give carbon dioxide along with the release of heat and light. The chemical equation to represent the burning of carbon is as follows:

Similarly, most carbon compounds burn in oxygen to give carbon dioxide with the evolution of heat and light. For example, hydrocarbon such as methane gives out a large amount of heat energy when burnt. It is because of this reason that methane is used as a fuel in industries.
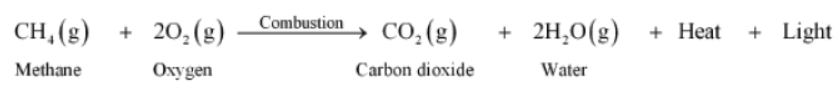
DO YOU KNOW?
The fuel that we use for cooking at home is Liquefied Petroleum Gas or LPG. LPG contains mainly butane and propane.
Do you know that burning of a carbon compound can help us in finding whether it is a saturated or an unsaturated compound?
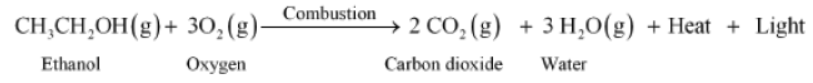
Alcohols also burn to give the same products as hydrocarbons. Ethyl alcohol or ethanol burns in air according to the given reaction.
Do you know?
Coal and petroleum were formed inside the Earth by the decomposition of organic matter. Coal is formed by the decomposition of trees and plants that lived millions of years ago, which got buried inside the Earth. Petroleum was formed by the decomposition of small plants and animals, which lived in the seas and oceans that got buried inside the seabed millions of years ago.
Kerosene and LPG stoves contain inlets for air to make sure that complete combustion takes place. Can you explain why?
This is because if the holes meant for the inlet of air are blocked or closed, then a yellow coloured sooty flame is produced. This shows that sufficient oxygen is not available for oxidation to take place and incomplete combustion is taking place.
Do you know that carbon compounds are oxidized on burning? Pure carbon is oxidized to give carbon dioxide when burnt in air. Similarly, hydrocarbons can be easily oxidized on burning. They are oxidized as they combine with oxygen (present in the air) on burning.
The gain of oxygen and loss of hydrogen is known as oxidation.
Substances that have the ability to oxidize other substances are known as oxidizing agents. Alkaline potassium permanganate and potassium dichromate are examples of oxidizing agents.
Compounds of carbon can also be oxidized partially with the help of oxidizing agents. For example, carboxylic acids are obtained when alcohols are treated with oxidizing agents such as alkaline potassium permanganate or acidified potassium dichromate.

In these reactions, alkaline potassium permanganate and acidified potassium dichromate act as oxidizing agents and convert alcohol to acids i.e., add oxygen to the starting material.
Activity 2:
Take around 3 mL of ethanol in a test tube. Warm it gently in a water bath. Add 5% solution of alkaline potassium permanganate drop by drop to the warm solution of ethanol and observe the colour change.
It will be observed that the pink colour of potassium permanganate solution disappears. This is because potassium permanganate is utilized for the oxidation of ethanol to produce ethanoic acid.
The chemical equation representing the oxidation of ethanol to ethanoic acid in the presence of potassium permanganate is as follows:

Addition and Substitution Reactions Of Carbon And Its Compounds
The compounds of carbon having double or triple bonds between the carbon atoms are known as unsaturated compounds, and the carbon compounds having single bonds between the carbon atoms are called saturated compounds. Do you know that unsaturated carbon compounds undergo addition reactions, while saturated compounds do not?
Addition reactions:
In an addition reaction, a substance is added to a hydrocarbon. A single product is obtained in this reaction.
In unsaturated hydrocarbons, double or triple bonds are present. Atoms of different elements like hydrogen, chlorine, bromine etc. are added to these compounds across the double or triple bonds using different reagents and catalysts.
Do You Know:
A catalyst is a substance that does not take part in a reaction, but increases the rate at which it proceeds. A catalyst is not affected during the course of a reaction and can be used repeatedly.
Hydrogen can be added to unsaturated hydrocarbons in the presence of nickel to yield saturated hydrocarbons. In this reaction, nickel acts as a catalyst.

This reaction is known as Hydrogenation reaction. It is used to obtain ghee from oil. Vegetable oil contains unsaturated fatty acids, and is liquid at room temperature. When they are hydrogenated, they become saturated and change to a solid state.
Do You Know:
Vegetable oil contains unsaturated fatty acids that are essential for our well-being, whereas saturated fatty acids like those present in ghee are not healthy. Therefore, vegetable oil should be preferred over ghee for cooking purposes
Important Note:
Saturated hydrocarbons do not undergo addition reactions. This is because there are no unsatisfied valencies in saturated compounds. Hence, we can differentiate saturated compounds from unsaturated compounds on the basis of addition reactions.
Do you know that saturated hydrocarbons are quite un-reactive? Also, they are inert to most reagents?
Low reactivity of saturated compounds is because of the presence of strong single bonds. However, in the presence of sunlight, the reaction between chlorine and alkanes takes place readily. This reaction is an example of substitution reaction.
Substitution reactions:
A substitution reaction is the one in which an atom or a group of atoms replaces another atom present in a molecule undergoing the reaction.
For example, chlorine replaces hydrogen from methane in the presence of sunlight.

Similar reactions in which bromine or iodine atoms replace hydrogen atoms are called bromination and iodination reactions respectively.
Properties and Reactions of Ethanol
Do you know that most substances used by us daily are compounds of carbon?
Substances like plastic, paper, cloth; fuels like petrol, diesel, and LPG (Liquefied petroleum Gas, which is used for cooking in our homes) are all compounds of carbon.
Therefore, it is important for us to know about some compounds of carbon that we use in our daily life. Here, we will discuss the physical and chemical properties of two important compounds of carbon i.e., ethanol and ethanoic acid. These two chemicals are very important commercially. Ethanol is present in all alcoholic beverages, and is also an important constituent of many medicines. Ethanoic acid is commonly used in homes as vinegar.
Ethanol or Ethyl alcohol, CH3CH2OH
Ethanol is commonly known as alcohol. It is an active ingredient of all alcoholic beverages, namely beer, wine, whisky, rum etc. It is also present in digestive and cough syrups. It is a neutral compound and has no effect on litmus paper. In industries, ethanol is produced by the fermentation of sugar. Intake of even a small quantity of pure ethanol can be lethal. If ethanol is consumed regularly for a very long time, then it can lead to many health problems and can also prove fatal.
Do You Know:
Ethanol is usually obtained from sugarcane. Sugarcane contains a large amount of sucrose. When molasses (a concentrated and dark liquid formed by boiling sugarcane juice) is fermented, ethanol is obtained. Fermentation is a metabolic process in which an organism converts a carbohydrate, such as starch or a sugar, into an alcohol or an acid. There are three major steps in production of ethanol by fermentation of molasses.
- Molasses are diluted with the help of water so that the solution contains about 10% sugar.
- Yeast is added to this solution and the temperature is maintained between 308 K to 313 K.
- The fermented matter which is filtered contains about 95% alcohol.
Physical properties:
1. It is a colourless liquid at room temperature, having a pleasant odour.
2. It is completely miscible with water in all proportions.
3. It is a very good organic solvent. Therefore, it is used in a lot of medicines, syrups, and tonics.
Chemical properties:
1. Reaction with sodium: Ethanol reacts with sodium to produce hydrogen gas.
Sodium ethoxide is also obtained in the reaction.
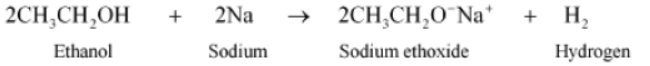
2. Dehydration reaction: When ethanol is heated with concentrated sulphuric acid, it loses one water molecule and gives ethene as a product.
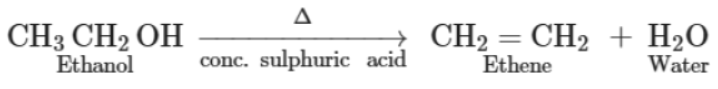
This reaction is known as dehydration reaction. In a chemical reaction, a substance is said to be dehydrated if it loses one or more water molecules. In this reaction, concentrated sulphuric acid acts as a dehydrating agent and removes water molecules from ethanol.
Combustion reaction: Combustion of alcohol results in the formation of carbon dioxide and water along with a large amount of heat and light energy.
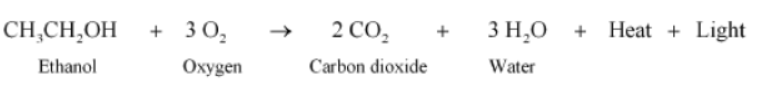
Do You Know:
As a large amount of energy is obtained when alcohol burns, it is used as an additive to petrol in some countries.
It burns to give only carbon dioxide and water. Thus, it is a clean fuel. It does not give out poisonous gases such as sulphur dioxide and nitrogen dioxide. Coal and petroleum contain nitrogen and sulphur, which cause pollution. Hence, coal and the fuels obtained from petroleum are not clean fuels.
Denatured alcohol:
To prevent the misuse of ethanol produced for industrial use, it is made unfit for drinking purpose by adding small amount of poisonous substances like methanol, pyridine and copper sulphate etc., to it. This is called denatured alcohol.
Harmful effects of drinking alcohol:
1) It slows down the activity of the nervous system as well as brain.
2) On a particular occasion, it leads to staggered movement, slurred speech etc.
3) It leads to blurred vision, dizziness and vomiting.
4) Drinking of large quantities of alcohol makes a man unconscious.
5) It can increase your risk of developing liver disease.
Heavy drinking makes a person addicted due to which it leads to a condition of financially bankrupt.
6) It can contribute to the condition that cause diabetes.
7) It can increase your risk of developing oral cancer.
8) Drinking of adulterated alcohol containing pyridine, causes severe poisoning leading to even death.
Properties and Reactions of Ethanoic Acid
Carboxylic acids are carbon compounds containing carboxyl functional group −COOH. The carboxyl group is named so because it consists of a carbonyl group attached to a hydroxyl group. Examples of carboxylic acid include methanoic acid (HCOOH), ethanoic acid (CH3COOH), propanoic acid (C2H5COOH), etc.
Classification of Carboxylic Acids
- On the basis of number of −COOH groups
- One −COOH group: Monocarboxylic acid, for example, HCOOH, CH3COOH, etc.
- Two −COOH groups: Dicarboxylic acid, for example, HOOC−COOH, etc.
Now, let us discuss an industrially important carboxylic acid.
Acetic Acid
Acetic acid is the common name of ethanoic acid (CH3COOH). Its dilute solution in water is known as vinegar, which is used for preserving food. Only 5-8 % solution of ethanoic acid is called vinegar, which is used as a preservative in pickles and salads. It is a weak acid in nature and turns blue litmus paper red.
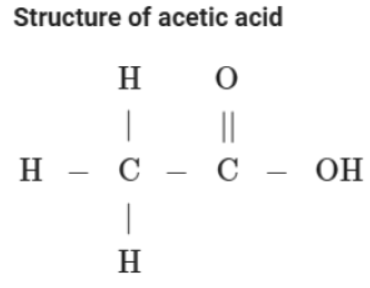
Preparation of acetic acid
Laboratory preparation: By oxidation of ethanol or ethanal (acetaldehyde) using acidified potassium dichromate solution
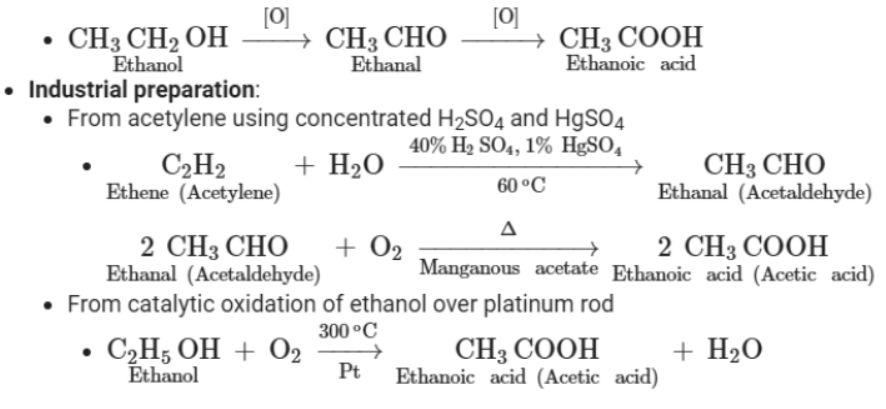
Physical properties
- It is a colourless, pungent smelling liquid.
- The melting point of ethanoic acid is 290 K. This is below room temperature. Thus, it freezes during winters. It looks similar to ice when it freezes. Therefore, it is also known as glacial acetic acid.
- It boils at a temperature of 118 oC.
- It is miscible with water, alcohol and ether in all proportions. It is hygroscopic in nature.
Chemical properties
- Acidic nature: Acetic acid is a weak acid. The following reactions prove the acidic nature of acidic acid.
- It turns blue litmus red.
- It reacts with active metals such as Zn and Mg to evolve hydrogen gas.
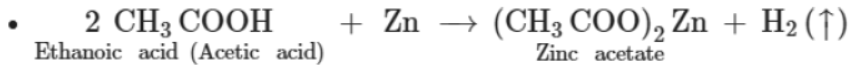
Reaction with alcohol or esterification reaction: The reaction of a carboxylic acid with an alcohol to form an ester is known as esterification reaction.
Curiosity Corner
Esters are sweet smelling organic compounds. As esters are volatile and pleasant to smell, they are generally used for making perfumes. They are also used as flavouring agents because of their fruity smell.
When ethanoic acid reacts with ethanol in the presence of an acid, ethyl ethanoate is formed.

Esters react in the presence of an acid or a base to give back alcohol and sodium salt of carboxylic acid as:

This reaction is used in the preparation of soaps and is known as saponification reaction.
- Reaction with a base:
We know that an acid and a base react to form salt and water. Similarly, ethanoic acid reacts with sodium hydroxide to form a salt, sodium ethanoate, and water. Sodium ethanoate is commonly known as sodium acetate.

- Reaction with carbonates and hydrogen carbonate:
Carbonates and bicarbonates are also basic in nature and react with ethanoic acid to form salt and water. Carbon dioxide is also formed in the reaction. The chemical equations involved are given as:


Sodium ethanoate is produced in the reaction. It is commonly known as sodium acetate.
- Reaction with phosphorous pentachloride
Acetic acid reacts with phosphorous chloride to form an acid derivative i.e. acetylchloride.

- Reaction with phosphorous pentoxide
Acetic acid forms another acid derivative when heated with phosphorous pentoxide. The acid derivative is acetic acid anhydride formed after removal of a water molecule from acid by phosphorous pentoxide.

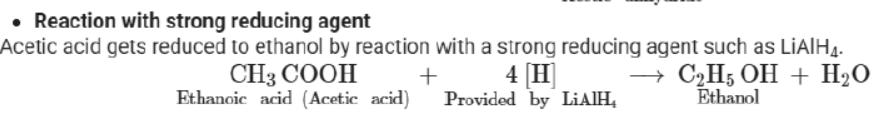
Tests for Acetic acid

Uses of acetic acid
Some uses of acetic acid are:
- In the manufacture of vinegar for preserving foods
- In the manufacture of cellulose acetate for photographic film
- As organic solvent
- In the production of polyethylene terephthalate used in soft drink bottles
- In the manufacture of polyvinyl acetate for synthetic fibres and fabric
Cleansing Action of Soaps & Detergents
We use soaps and detergents in our homes for almost all cleaning purposes. What are soaps and detergents? Soaps are sodium or potassium salts of higher fatty acids such as oleic acid (C17H33COOH), stearic acid (C17H35COOH), palimitic acid (C15H31COOH), etc. On the other hand, detergents are salts of higher sulphonic acids such as dedecylbenzenesulphonate (C18H30SO3Na).
Preparation of Soaps and Detergents:
Soaps are prepared by gentle heating of oil or fat with sodium hydroxide solution till it boils. After few minutes of boiling sodium chloride is added to the solution to separate out soap. The solution is cooled and soap is obtained at the top of the solution. To obtain glycerol which is a byproduct of the formation of soap is obtained by hydrolyzing oil in presence of bases such as potassium hydroxide.
Oil + NaOH → Soap + Glycerol
This process is known as saponification.
Similarly, the long chain hydrocarbons obtained after refining of petroleum when treated with concentrated sulphuric acid and neutralized with sodium hydroxide produce detergents.
Do you know that soaps and detergents help in dissolving dirt (oil and grease) in water? A soap solution or detergent makes dirt soluble in water, thereby cleaning a dirty cloth.
To understand the cleansing action of soap, we first have to understand its structure. Soap has one polar end (the end with sodium or potassium ion) and one non-polar end (the end with fatty acid chain) as shown in the figure. The polar end is hydrophilic in nature i.e., this end is attracted towards water. The non-polar end is hydrophobic, but lypophilic in nature i.e., it is attracted towards hydrocarbons, but not towards water.
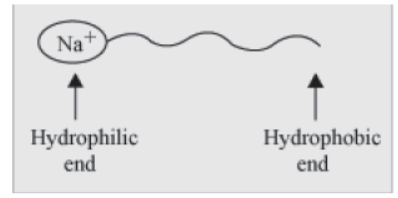
A soap molecule
Do you know what happens when soap molecules are added to water? When soap molecules are present in water, the molecules arrange themselves in the form of a cluster in such a manner that their hydrophobic ends are away from the water molecules and their hydrophilic or ionic ends are towards the water molecules (as shown in the following figure). This is known as micelle formation and the cluster that is formed is called a micelle.
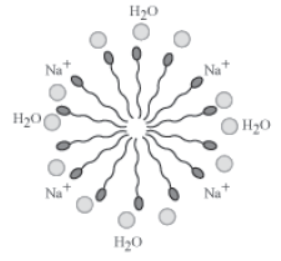
Micelle formation
We know that soaps and detergents make oil and dirt present in a cloth dissolve in water, thereby making the cloth clean.
DO YOU KNOW?
Soap does not work properly when water is hard. This is primarily because hard water contains salts of calcium and magnesium. When soap is added to hard water, it reacts with these salts to form an insoluble substance called scum. A lot of soap is wasted in the process. In addition, soaps do not give lather with hard water. When formed, the scum sticks to the cloth and decreases the cleansing ability of the soap.
This problem is overcome by using detergents.
Advantages of Detergents over soaps:
Detergents clean efficiently in hard water whereas soaps are rendered inactive in hard water.
- Detergents can work well in acidic medium, whereas soaps do not work in an acidic medium.
However, as detergents are non – biodegradable they cause pollution of water and soil. This is one of the major drawbacks of using detergents.


