Please refer to Assignments Class 10 Science Chemical Reactions and Equations Chapter 1 with solved questions and answers. We have provided Class 10 Science Assignments for all chapters on our website. These problems and solutions for Chapter 1 Chemical Reactions and Equations Class 10 Science have been prepared as per the latest syllabus and books issued for the current academic year. Learn these solved important questions to get more marks in your class tests and examinations.
Chemical Reactions and Equations Assignments Class 10 Science
Very Short Answer Questions
Question. Balance the following chemical equation:
Pb(NO3)2 → PbO + NO2 + O2
Answer : 2Pb(NO3)2 →Heat 2PbO + 4NO2 + O2
Question. Identity the type of reaction in the following example:
Na2SO4(aq) + BaCl2(aq) → BaSO4(s) + 2NaCl(ag)
Answer : Double displacement reaction.
Question. Is burning of a candle wax a physical or a chemical change?
Answer : Burning of a candle wax is a chemical change.
Question. Write a balanced equation for the chemical reaction that can be characterised as precipitation reaction.
Answer : AgNO3(aq) + NaCl(aq) → AgCl(s) + NaNO3(aq)
It is a precipitation reaction.
Question. What happens chemically when quick lime is added to water filled in a bucket?
Answer : Calcium hydroxide (Slaked lime) is formed with evolution of heat and hissing sound.
CaO(s) + H2O(l) → Ca(OH)2(aq)
Question. Define oxidation and reduction.
Answer : Oxidation is a process in which oxygen is added or loss of electrons take place. Reduction is a process in which hydrogen is added or gain of electrons takes place.
Question. Write a complete balanced chemical equation for the following reaction: Sodium hydroxide + Sulphuric acid →
Answer : 2NaOH + H2SO4 → Na2SO4 + 2H2O
Question. Name and state the law which is kept in mind when we balance chemical equations.
Answer : Law of conservation of mass. It states Matter can neither be created nor be destroyed. The total mass of reactants must be equal to total mass of products.
Question. Give an example of double displacement reaction
(only with complete balanced equation).
Answer : BaCl2(aq) + H2SO4(dil.) → BaSO4(s) + 2HCl(aq)
It is a double displacement reaction.
Question. In electrolysis of water, why is the volume of gas collected over one electrode double that of gas collected over the other electrode.
Answer : Water contains hydrogen and oxygen in the ratio of 2 : 1, that is why volume of H2 is double than that of oxygen.
Question. What can be seen when a strip of copper metal is placed in a solution of silver nitrate?
Answer : The solution will become blue, shiny silver metal will get deposited.
Cu(s) + 2AgNO3(aq) → Cu(NO3)2 (aq) + 2Ag(s)
Question. Write a balanced chemical equation for the reaction between sodium chloride and silver nitrate indicating the physical state of the reactants and the products.
Answer : (Silver Nitrate)
(Img 2)
Question. Complete and balance the following equation:
Fe2O3 + Al →
Answer : Fe2O3 + 2Al → 2Fe + Al2O3
Question. Balance the given chemical equation:
Al(s) + CuCl2(aq) → AlCl3(aq) + Cu(s)
Answer : 2Al(s) + 3CuCl2(aq) → 2AlCl3(aq) + 3Cu(s)
Question. N2(g) + 3H2(g) → 2NH3(g), name the type of reaction.
Answer : Combination reaction.
Question. If copper metal is heated over a flame it develops a coating. What is the colour and composition of coating?
Answer : Black coloured coating is formed. It is a due to formation of copper oxide.
Question. Balance the given chemical equation:
FeSO4(s) →Heat Fe2O3(s) + SO2(g) + SO3(g)
Answer : 2FeSO4(s) →Heat Fe2O3(s) + SO2(g) + SO3(g)
Short Answer Questions
Question. Write balanced chemical equation for the following reactions:
a. Hydrogen sulphide burns in air to give water and sulphur dioxide.
b. Barium chloride reacts in aqueous solution with zinc sulphate to give zinc chloride and barium sulphate.
Answer :
a. 2H2S(g) + 3O2(g) → 2H2O(l) + 2SO2(g)
b. BaCl(aq) + ZnSO(aq) → BaSO4 (s) + ZnCl2(aq)
Question. List two observations that are noticed when an iron nail is put inside copper sulphate solution. Write the chemical equation for the reaction that occurs.
Answer : The blue coloured solution will become pale green.
Reddish brown metal will get deposited.
Fe(s) + CuSO (aq) → FeSO (aq) + Cu(s)
(Blue) (Pale Green) (Reddish brown)
Question. Name the reducing agent in the following reaction:
3MnO2 + 4A1 → 3Mn + Al2O3
State which is more reactive, Mn or Al and Why?
Answer : Al is the reducing agent. Al is more reactive than Mn.
Reason: It is because Al is displacing Mn from MnO2.
Question. “We need to balance a skeleton chemical equation”.
Give reason to justify the statement.
Answer : We must balance a skeletal equation so as to ensure that the reaction follows ‘Law of conservation of mass’.
The total mass of reactants must be equal to the total mass of products, that is why all reactions should he balanced.
Question. A metal is treated with dilute H2SO4. The gas evolved is collected by the method as shown in figure.
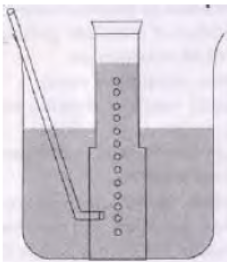
Answer the following questions:
a. Name the gas liberated.
b. Name the method used for collection of gas.
c. Is the gas soluble or insoluble in water?
d. Is the gas lighter or heavier than air?
Answer :
a. The gas liberated is H2.
b. It is collected by downward displacement of water.
c. The gas is insoluble in water.
d. Hydrogen gas is lighter than air.
Question. What is a redox reaction? Identify the substances oxidised and the substance reduced in the following reactions:
a. MnO2+ 4HCl → MnCl2 + Cl2 + H2O
b. CuO + H2 → Cu + H2O
Answer : Redox reaction is a reaction in which oxidation and reduction takes place simultaneously.
c. HCl is the substance oxidised, MnO2 is the substance getting reduced.
d. H2 is getting oxidised, CuO is getting reduced.
Question. Consider the following chemical equation:
X + Barium chloride → Y + Sodium chloride
(White ppt)
Identify (a) X and Y (b) The type of reaction.
Answer : (a) X is silver nitrate, Y is silver chloride.
2AgNO (aq) + BaCl (aq) → 2AgCl + Ba(3NO3)2 (aq)
(White ppt)
(b) The reaction is an example of double displacement (precipitation) reaction.
Question. (a) Write a balanced chemical equation for the process of photosynthesis.
(b) When do desert plant take up carbon dioxide and perform photosynthesis.
Answer : a. (Img 3)
b. Desert plant take up carbon dioxide and perform photosynthesis at night.
Question. On heating copper powder in air, the surface of copper powder becomes coated with black CuO. How can this black coating be converted into brown copper? Write chemical equation for the reaction that occurs during the colour change.
Answer : 2Cu + O2 →Heat 2CuO
(Black)
Copper oxide on heating with H2 will change back to reddish brown copper metal.
CuO(s) + H2(g) → Heat Cu(s) + H2O(g)
Question. Translate the following statements into chemical equations and then balance them:
a. Hydrogen gas combines with nitrogen to form ammonia.
b. Hydrogen sulphide gas burns in air to give water and sulphur dioxide.
c. Barium chloride reacts with aluminium sulphate to give aluminium chloride and a precipitate of barium sulphate. State the two types in which this reaction can be classified.
d. Potassium reacts with water to give potassium hydroxide and hydrogen gas.
Answer :
a. 3H2(g) + N2(g) → 2NH3(g)
b. 2H2S(g) + 3O2(g) → 2H2O(l) + 2SO2(g)
c. 3BaCl2(aq) + Al2(SO4)3(aq) → 3BaSO4(s) + 2AlCl3(aq)
This reaction can be classified as a double displacement reaction as the two reacting species (BaSO4, Al2SO4)3 undergoes mutual exchange of ions. It can also be classified as precipitation reaction, since a white ppt. of BaSO4 is obtained.
d. 2K(s) + 2H2O(l) → 2KOH(aq) + H2(g)
Question. Write the essential condition for the following reaction to take place:
2AgBr → 2Ag + Br2
Write application of this reaction.
Answer : The reaction will take place in presence of sunlight.
This reaction is used in black and white photography.
Question. What is observed when a solution of potassium iodide is added to a solution of lead nitrate? Name the type of reaction. Write a balanced chemical equation to represent the above chemical reaction.
Answer : Yellow precipitate is formed due to formation of lead iodide.
It is a precipitation as well as double displacement reaction.
Pb(NO ) (aq) + 2KI (ag) → PbI (s) + 2KNO (aq)
Yellow ppt
Question. Using a suitable chemical equation justify that some chemical reactions are determined by
a. Change in colour.
b. Change in temperature.
Answer :
a. Change in colour: Blue coloured crystalline copper sulphate crystals changes to white CuSO4 (anhydrous) on heating.
CuSO4.5H2O →Heat CuSO4 + 5H2O
(Blue) (Dirty white)
Question. Write a balanced chemical equation for the process of photosynthesis and the conditions of reaction giving physical state of all substances.
Answer : 6CO2 (g) 6H2O(l) → sunlight C6H12O6 (aq) + 6O2 (g)
Question. Translate the following statement into chemical equation and then balance it. “A metal in the form of ribbon burns with a dazzling white flame and changes into white powder.”
Answer : 2Mg(s) + O2(g) →Burning 2MgO(s) + Light + Heat
Question. Why does the colour of copper sulphate solution changes when an iron pin is dipped in it?
Answer :
Iron being more reactive displaces copper from copper sulphate (Blue) solution to form iron (II) sulphate (Pale green) solution and reddish brown copper metal
gets deposited.
Fe(s) + CuSO4 → FeSO4(aq) + Cu(s)
Question. It has been found that marble of Taj is getting corroded due to development of industrial areas around it.
Explain this fact giving a chemical equation.
Answer : Taj is made up of CaCO3 which reacts with acid formed by pollution of SO2(g) and NO2 from Mathura refinery and other industries.
CaCO3 + 2 H2SO4 → CaSO4 + CO2 + 2H2O
CaCO3 + 2HNO3 → Ca(NO3)2 + H2O + CO2
Question. When a solution of potassium iodide is added to a solution of lead nitrate in a test tube, a reaction takes place.
a. What type of reaction is this?
b. Write the balanced chemical equation to represent the above reaction.
Answer :
a. It is precipitation as well as double displacement reaction.
b. Pb(NO3) (aq) + 2KI (aq) → Pbl2(s) + 2KNO3(aq)
Question. Consider the chemical equations given below and answer the questions which follow:
(i) CuO + H2 →Heat Cu + H2O
(ii) ZnO + C →Heat Zn + CO
a. Name the substances that are oxidised and
reduced respectively in each case,
b. Identify the reducing agent in each case.
Answer :
a. H2 is getting oxidised to H2O, CuO getting reduced to Cu.
b. H2 is reducing agent in (i) reaction whereas C is reducing agent in second reaction.
Question. Identify the type of each of the following reactions:
a. A reaction in which a single product is formed from two or more reactants.
b. The reaction mixture becomes warm.
c. An insoluble substance is formed.
d. External surface of the container in which reaction takes place becomes cold.
Answer :
a. Combination reaction
b. Exothermic reaction
c. Precipitation reaction (Double displacement reaction)
d. Endothermic reaction
Question. (a) Complete the following equation for the chemical reaction:
FeSO4(s) Heat Fe2O3 + ___ +___
(b) What happens when water is added to quicklime (CaO)? Write the chemical equation.
Answer : (a) 2FeSO4(s) →Heat Fe2O3(s) + SO2(g) + SO3(g)
(b) Slaked lime Ca(OH)2 is formed. Hissing sound and lot of heat is also produced:
CaO(s) + H2O(l) → Ca(OH)2(aq)
Question. Write balanced chemical equations for the following reactions:
a. Silver bromide on exposure to sunlight decomposes into silver and bromine.
b. Sodium metal reacts with water to form sodium hydroxide and hydrogen gas.
Answer : a. 2AgBr(s) sunlight 2Ag(s)+ Br2(g)
b. 2Na(s) + 2H2O(l) → 2NaOH(l) + H2(g)
Question. Identify the type of reaction from the following equations:
a. CH4 + 2O2 → CO2 + 2H2O
b. Pb(NO3)2 + 2KI → PbI2 + 2KNO3
c. CaO + H2O → Ca(OH)2
d. CuSO4 + Zn → ZnSO4 + Cu
Answer :
a. Oxidation reaction
b. Double displacement reaction
c. Combination reaction
d. Displacement reaction
Question. Write balanced equation for the reaction between Mg and hydrochloric acid. Name the product obtained, identify the type of reaction.
Answer :
Mg(s) + 2HCl(dil) → MgCl2(aq) + H2(g)
Magnesium chloride and hydrogen gas are formed in this reaction. It is a displacement reaction.
Question. a. What is colour of ferrous sulphate crystals? How does this colour change after heating?
b. Name the products formed on strongly heating ferrous sulphate crystals. What type of chemical reaction occurs in this change?
Answer :
a. FeSO4 7H2O → crystals are pale green in colour.
They become dirty white on heating.
b. Ferric oxide, sulphur dioxide and sulphur trioxide are formed:
2FeSO4(s) →HeatFe2O3(s) + SO2(g) + SO3(g)
It is a decomposition reaction.
Question. Reaction of compound X with aluminium is used to join railway tracks or cracked machine parts.
a. Identify the compound.
b. Name the reaction.
c. Write a balanced chemical equation for the reaction.
Answer :
a. The compound X is Fe2O3 (Ferric oxide) or Iron (III) oxide.
b. It is called Thermite Reaction.
c. 2Al(s) + Fe2O3(s) →Heat Al2O3(s) + 2Fe(l)
Question. A white salt on heating decomposes to give brown fumes and a white residue is left behind.
a. Name the salt.
b. Write the equation for the decomposition reaction.
Answer :
a. Lead nitrate
b. 2Pb(NO3)3 (s) → 2PbO(s) + 4NO2 (g) + O2 (g)
(White) (Brown)
Question. Giving an example list two important information which makes a chemical equation more useful (informative).
Answer :
a. It should include physical states of reactants and products.
b. It should specify conditions under which reaction takes place e.g.,
2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g)
(Cold)
H2(g) + Cl2(g) sunlight 2HCl(g)
Question. Using balanced chemical equation explain the difference between a displacement reaction and a double displacement reaction.
Answer :
Displacement reaction: A reaction in which a more reactive element displaces a less reactive element from its salt solution e.g.,
2KBr(aq) + Cl2(g) → 2KCl(aq) + Br2(aq)
Double displacement reaction: A reaction in which two compounds exchange their ions to form two new compounds e.g.,
KOH + HNO3 → KNO3 + H2O
Question. Give one example of each:
a. Chemical reaction showing evolution of a gas.
b. Change in colour of a substance during chemical reaction.
Answer :
a. 2K(s) + 2H2O(l) → 2KOH(aq) + H2(g)
b. FeSO4.7H2O(aq) → Heat FeSO4(s) + 7H2O(l)
(Pale green) (Dirty white)
Question. Give an example each for thermal decomposition and photochemical decomposition reactions. Write balanced chemical equation also.
Answer :
Thermal decomposition:
ZnCO3(s) →Heat ZnO(s) + CO2(g)
Photochemical decomposition:
2AgI(s) →sunlight 2Ag(s) + I2(g)
Question. Why are decomposition reactions called the opposite
of combination reactions? Give chemical equations for
these reactions.
Answer :
In decomposition reactions, a compound is broken down into one or more elements or compounds e.g.,
CaCO3(s) →Heat CaO(s) + CO2(g)
In combination reactions, two or more elements or compounds combine to form a new compound.
Therefore, decomposition reactions are opposite to combination reactions e.g.,
3Mg(s) + N2(g) →Burns Mg3N2(s)
Question. When a metal X is added to salt solution of metal Y, the following chemical reaction takes place:
Metal X + Salt solution of Y →
Salt solution of X + Metal Y
Mention the inference you draw regarding the reactivity of metal X and Y and also the type of reaction. State the reason of your conclusions.
Answer :
X is more reactive than Y because , X displaces Y from its salt solution. Hence, this reaction is an example of displacement reaction.
Question. When magnesium ribbon bums in air or oxygen, a product is formed. State the type of chemical reaction and name the product formed in the reaction. Write balanced chemical equation for the reaction.
Answer :
It is a combination reaction. The product formed is magnesium oxide:
2Mg(s) + O2(g) → 2MgO(s)
Long Answer Questions
Question. Write balanced chemical equation for the following statements:
a. NaOH solution is heated with zinc granules.
b. Excess of carbon dioxide is passed through lime water.
c. Dilute sulphuric acid is added to sodium carbonate.
d. Egg shell is dropped in hydrochloric acid,
e. Copper (II) oxide reacts with dilute hydrochloric acid.
Answer :
a. Zn(s) + 2NaOH → Na2ZnO2 + H2
b. Ca(OH)2 + 2CO2 → Ca(HCO3)2
c. Na2CO3 + H2SO4 → Na2SO4 + H2O + CO2
d. CaCO3(s) + 2HCl(dil) → CaCl2 + H2O + CO2
e. CuO(s) + 2HC1 → CuCl2 + H2O
Question. Identify the type of chemical reaction in the following statement and define each of them:
a. Digestion of food in our body.
b. Rusting of iron.
c. Heating of manganese dioxide with aluminium powder.
d. Blue colour of copper sulphate solution disappears when iron filings are added to it.
e. Dilute hydrochloric acid is added to sodium hydroxide solution to form sodium chloride and water.
Answer :
a. Decomposition reaction: It is a process in which a compound is broken down into simple substances.
b. Oxidation: The process in which oxygen is added or electrons are lost.
c. Displacement reaction: The reaction in which a more reactive element can displace a less reactive element from its salt solution. Oxidation and Reduction are taking place simultaneously in rusting of iron.
d. Displacement reaction: The reaction in which a more reactive element can displace a less reactive element.
e. Neutralisation reaction: The reaction in which acid reacts with base to form salt and water.
Question. Define chemical reaction. State four observations which helps to determine whether a chemical reaction has taken place or not. Write one example of each observation with a balanced chemical equation.
Answer :
Chemical reaction is a reaction which represents a chemical change.
a. Change in colour:
Cu(s) + 2AgNO (aq) → Cu(NO3) (aq) + 2Ag(s)
(Colourless) (Blue)
b. Evolution of gas:
CaCO3 (s) →Heat CaO(s) + CO2(g)
Carbon dioxide gas
c. Formation of precipitate:
Pb(NO3)2 (aq) + 2KI PbI (s) → 2KNO3 (aq)
(Yellow ppt)
d. Change in temperature:
CaO(s) + H2O(l) → Ca(OH)2 + Heat


