Please refer to Assignments Class 10 Science Carbon and Its Compound Chapter 4 with solved questions and answers. We have provided Class 10 Science Assignments for all chapters on our website. These problems and solutions for Chapter 4 Carbon and Its Compound Class 10 Science have been prepared as per the latest syllabus and books issued for the current academic year. Learn these solved important questions to get more marks in your class tests and examinations.
Carbon and Its Compound Assignments Class 10 Science
Very Short Answer Type Question
Question. Draw the structure of butanone molecule, CH3COC2H5.
Answer:
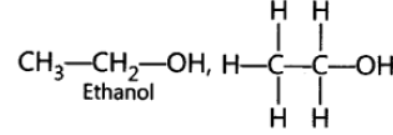
Question. Write the name and formula of the 2nd member of the series of carbon compounds whose general formula is CnH2n+1OH
Answer: Ethanol, C2H5OH or CH3CH2OH
Question. Write the name and formula of the 2nd member of the series of carbon compounds whose general formula is CnH2n.
Answer:
C3H6, H2C=CH—CH3
Propene is second member of series whose general formula is CnH2n.
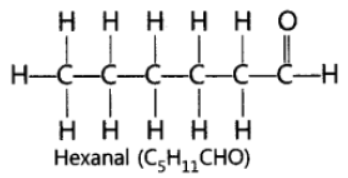
Question. Draw the structure of the hexanal molecule, C5H11CHO.
Answer:
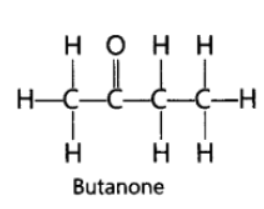
Question. Butanone is a four carbon per molecule compound. Name the functional group present in it.
Answer: Ketone
Question. 30 Name the following compound: img
Answer: 1-Hexyne is IUPAC name of the compound
Question. Name the functional group present in each of the following organic compounds:
(i) C2H5CI
(ii) C2H5OH
Answer:
(i) (—Cl) Halogen (Chloro)
(ii) (—OH) Alcohol
Question. Name the functional group present in each of the following compounds:
(i) HCOOH
(ii) C2H5CHO
Answer:
(i) —COOH (Carboxylic acid)
(ii) —CHO (Aldehyde)
Question. Name the functional group present in each of the following organic compounds:
(i) CH3COCH3
(ii) C2H5COOH
Answer:

Question. Write the name and formula of the second member of the carbon compounds having functional group —OH.
Answer:
Question. Write the name and formula of the first member of the carbon compounds having functional group —CHO.
Answer:
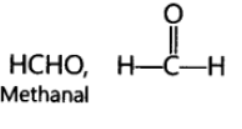
Question. What change will you observe if you test soap with litmus paper (red and blue)?
Answer. Since soap is basic in nature, it will turn red litmus blue. However, the colour of blue litmus will remain blue.
Question. Why do ionic compounds have high melting points?
Answer. Ionic compounds have strong electrostatic forces of attraction between the ions. Therefore, it requires a lot of energy to overcome these forces. That is why ionic compounds have high melting points.
Question. Would you be able to check if water is hard by using a detergent?
Answer. Detergents are ammonium or sulphonate salts of long chain carboxylic acids. Unlike soap, they do not react with calcium and magnesium ions present in hard water to form scum. They give a good amount of lather irrespective of whether the water is hard or soft. This means that detergents can be used in both soft and hard water. Therefore, it cannot be used to check whether the water is hard or not.
Question. People use a variety of methods to wash clothes. Usually after adding the soap, they ‘beat’ the clothes on a stone, or beat it with a paddle, scrub with a brush or the mixture is agitated in a washing machine. Why is agitation necessary to get clean clothes?
Answer. A soap molecule has two parts namely hydrophobic and hydrophilic. With the help of these, it attaches to the grease or dirt particle and forms a cluster called micelle. These micelles remain suspended as a colloid. To remove these micelles (entrapping the dirt), it is necessary to agitate clothes.
Question. Give a test that can be used to differentiate chemically between butter and cooking oil.
Answer. Butter contains saturated fats. Therefore, it cannot be hydrogenated. On the other hand, oil has unsaturated fats. That is why it can be hydrogenated to saturated fats (solids).
Question. What are oxidising agents?
Answer. Some substances such as alkaline potassium permanganate and acidified potassium dichromate are capable of adding oxygen to others. These are known as oxidising agents.
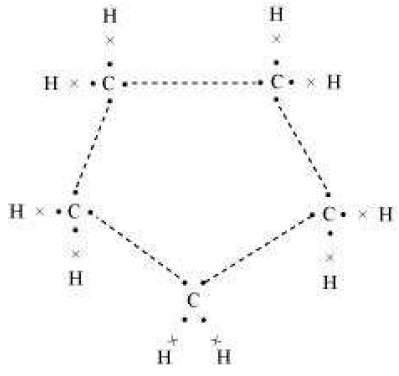
Question. What will be the formula and electron dot structure of cyclopentane?
Answer. The formula for cyclopentane is C5H10. Its electron dot structure is given below.
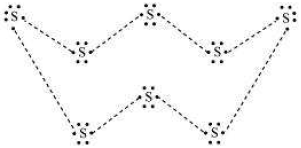
Question. What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulphur? (Hint − the eight atoms of sulphur are joined together in the form of a ring.)
Answer. Electron dot structure of a sulphur molecule
Question. What would be the electron dot structure of carbon dioxide which has the formula CO2?
Answer. Electron dot structure of CO2 is

Question. Why are carbon and its compounds used as fuels for most applications?
Answer. Most of the carbon compounds give a lot of heat and light when burnt in air. Saturated hydrocarbons burn with a clean flame and no smoke is produced. The carbon compounds, used as a fuel , have high calorific values. Therefore, carbon and its compounds are used as fuels for most applications.
Question. Which of the following hydrocarbons undergo addition reactions:
C2H6, C3H8, C3H6, C2H2 and CH4.
Answer. Unsaturated hydrocarbons undergo addition reactions. Being unsaturated hydrocarbons, C3H6 and C2H2 undergo addition reactions.
Question. Why is the conversion of ethanol to ethanoic acid an oxidation reaction?
Answer.

Since the conversion of ethanol to ethanoic acid involves the addition of oxygen to ethanol, it is an oxidation reaction.
Question. Give the names of the following functional groups:
(i) —OH (ii) —COOH
Answer: (i) Alcohol group (ii) Carboxylic acid group
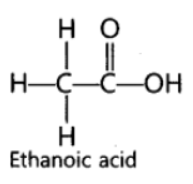
Question. Draw the structure of CH3COOH molecule.
Answer:
Question. Draw the structure of ethanol molecule.
Answer:

Short Answer Type Questions :
Question. What is an ‘esterification’ reaction? Describe an activity to show esterification.
Answer:
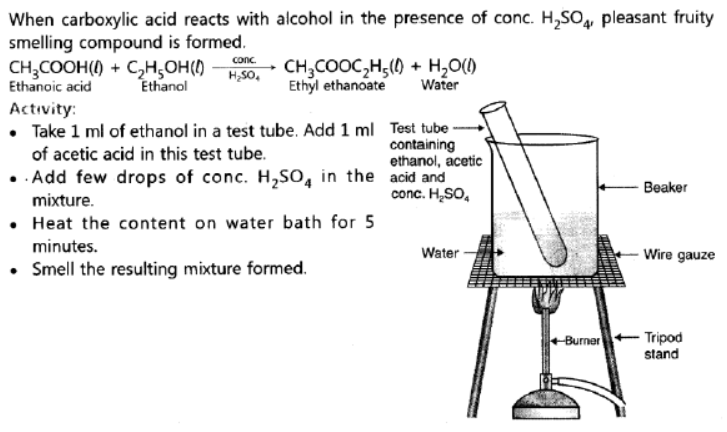
Question. What are isomers? Draw the structures of two isomers of butane, C4H10. Why can’t we have isomers of first three members of alkane series?
Answer: Those compounds, which have same molecular formula but different structural formulae are called isomers.
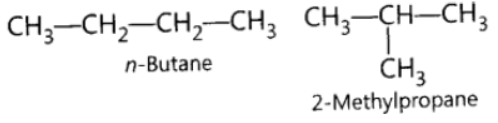
In first three members of alkane series, branching is not possible. Therefore, we cannot have isomers.
Question. How can ethanol and ethanoic acid be differentiated on the basis of their physical andchemical properties?
Answer. 1. Ethanol is a liquid at room temperature with a pleasant odour while ethanoic acid has vinegar-like smell. The melting point of ethanoic acid is 17°C. This is below room temperature and hence, it freezes during winters.
2. Ethanoic acid reacts with metal carbonates and metal hydrogencarbonates to form salt, water, and carbon dioxide gas while ethanol does not react with them.
Question. Explain the formation of scum when hard water is treated with soap.
Answer. Soap does not work properly when the water is hard. A soap is a sodium or potassium salt of long chain fatty acids. Hard water contains salts of calcium and magnesium. When soap is added to hard water, calcium and magnesium ions present in water displace sodium or potassium ions from the soap molecules forming an insoluble substance called scum. A lot of soap is wasted in the process.
Question. What are the two properties of carbon which lead to the huge number of carbon compounds we see around us?
Answer. The two features of carbon that give rise to a large number of compounds are as follows:
(i) Catenation − It is the ability to form bonds with other atoms of carbon.
(ii) Tetravalency − With the valency of four, carbon is capable of bonding with four other atoms.
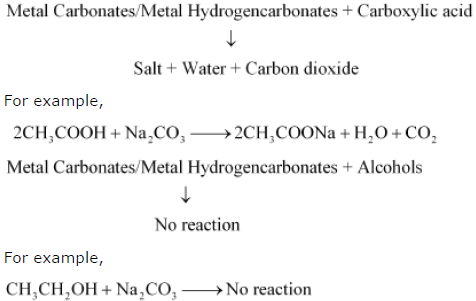
Question.How would you distinguish experimentally between an alcohol and a carboxylic acid?
Answer. We can distinguish between an alcohol and a carboxylic acid on the basis of their reaction with carbonates and hydrogen carbonates. Acid reacts with carbonate and hydrogen carbonate to evolve CO2 gas that turns lime water milky.
carbonate / Metal hydrogen carbonate + carboxylic acid
↓
Salt + Water + Carbon dioxide
Alcohols, on the other hand, do not react with carbonates and hydrogen carbonates.
Question. How many structural isomers can you draw for pentane?
Answer. Three structural isomers are possible for pentane.
(i) CH3 — CH2 — CH2 — CH2 — CH3
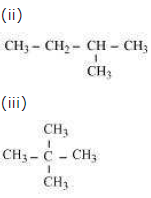
Question. In the electrolytic refining of a metal M, what would you take as the anode, the cathode and the electrolyte?
Answer. In the electrolytic refining of a metal M:
Anode → Impure metal M
Cathode → Thin strip of pure metal M
Electrolyte → Solution of salt of the metal M
Question. Explain the mechanism of the cleaning action of soaps.
Answer. Cleansing action of soaps:
The dirt present on clothes is organic in nature and insoluble in water. Therefore, it cannot be removed by only washing with water. When soap is dissolved in water, its hydrophobic ends attach themselves to the dirt and remove it from the cloth. Then, the molecules of soap arrange themselves in micelle formation and trap the dirt at the centre of the cluster. These micelles remain suspended in the water. Hence, the dust particles are easily rinsed away by water.

Question. What is a homologous series? Explain with an example.
Answer. A homologous series is a series of carbon compounds that have different numbers of carbon atoms but contain the same functional group.
For example, methane, ethane, propane, butane, etc. are all part of the alkane homologous series. The general formula of this series is CnH2n+2.
Methane CH4
Ethane CH3CH3
Propane CH3CH2CH3
Butane CH3CH2CH2CH3
It can be noticed that there is a difference of −CH2 unit between each successive compound.
Question. What is hydrogenation? What is its industrial application?
Answer. Hydrogenation is the process of addition of hydrogen. Unsaturated hydrocarbons are added with hydrogen in the presence of palladium and nickel catalysts to give saturated hydrocarbons.
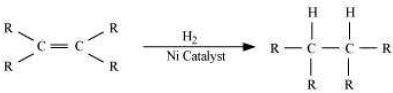
This reaction is applied in the hydrogenation of vegetables oils, which contain long chains of unsaturated carbons.
Question. A mixture of oxygen and ethyne is burnt for welding. Can you tell why a mixture of ethyne and air is not used?
Answer.
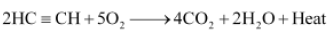
When ethyne is burnt in air, it gives a sooty flame. This is due to incomplete combustion caused by limited supply of air. However, if ethyne is burnt with oxygen, it gives a clean flame with temperature 3000°C because of complete combustion. This oxy-acetylene flame is used for welding. It is not possible to attain such a high temperature without mixing oxygen. This is the reason why a mixture of ethyne and air is not used.
Question. Define homologous series of organic compounds. List its two characteristics.
Write the name and formula of the first member of the series of alkenes.
Answer: The series of organic compounds having same functional group and similar chemical properties is called homologous series.
Each member differs from successive member by —CH2— group. The difference in molecular weight between two successive members is 14 u.
Characteristics:
(i) It has same general formula, from which, all members can be derived.
(ii) They have similar chemical properties.
C2H4, CH2=CH2, Ethene is first member of alkene series.
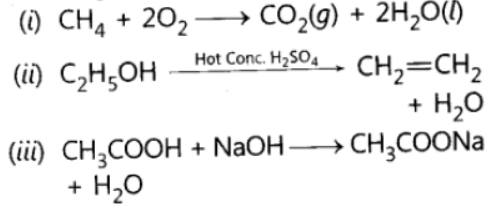
Question. 50 Complete the following equations:
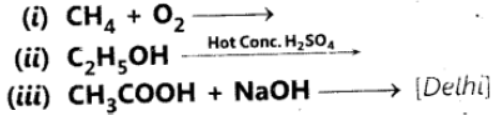
Answer:
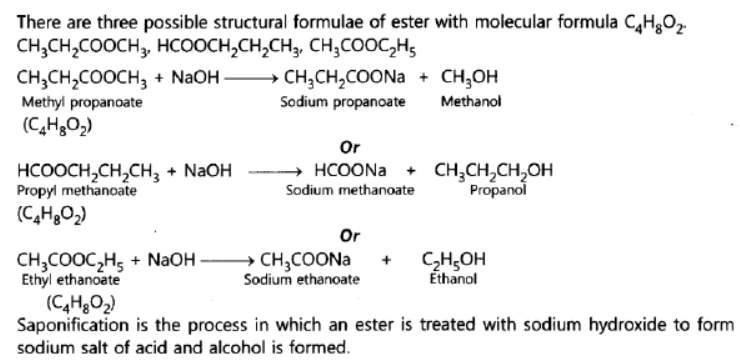
Question. An ester has the molecular formula C4H8O2. Write its structural formula. What happens when this ester is heated in the presence of sodium hydroxide solution? Write the balanced chemical equation for the reaction and name the products. What is a saponification reaction?
Answer:
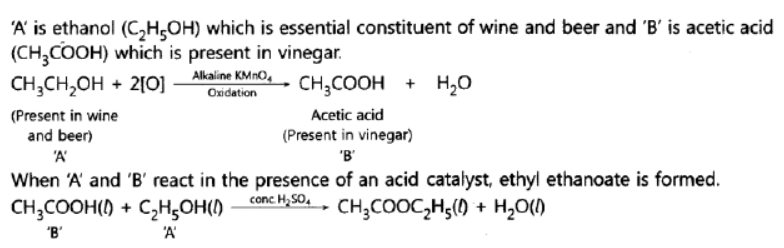
Question. An organic compound ‘A’ is an essential constituent of wine and beer. Oxidation of ‘A’ yields an organic acid ‘B’ which is present in vinegar. Name the compounds ‘A’ and ‘B’ and write their structural formula. What happens when ‘A’ and ‘B’ react in the presence of an acid catalyst? Write the chemical equation for the reaction.
Answer:
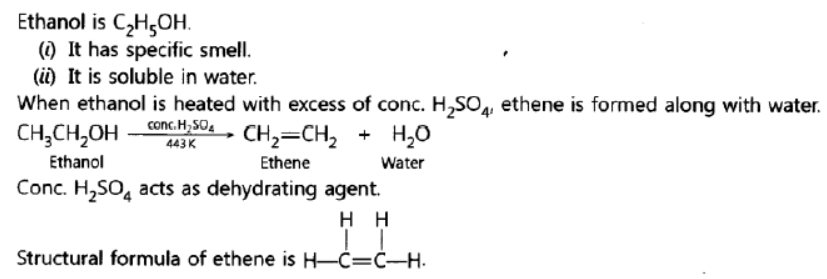
Question. What is ethanol? State its two properties. What happens when it is heated with excess of cone. H2SO4 at 443 K? What role does cone. H2SO4 play in this reaction? Write chemical equation of the reaction involved and the structural formula of the main product formed.
Answer:
Question. Why homologous series of carbon compounds are so called? Write chemical formula of two consecutive members of a homologous series and state the part of these compounds that determines their (i) physical properties, and (ii) chemical properties.
Answer: The series consists of members of same family with similar physical and chemical properties, therefore, called homologous series
(i) CH3OH, and (ii) CH3CH2OH are two consecutive members of homologous series.
Alkyl group —CH3 and —CH3CH2 part determines physical properties. Functional group —OH
determines chemical properties of the compounds.
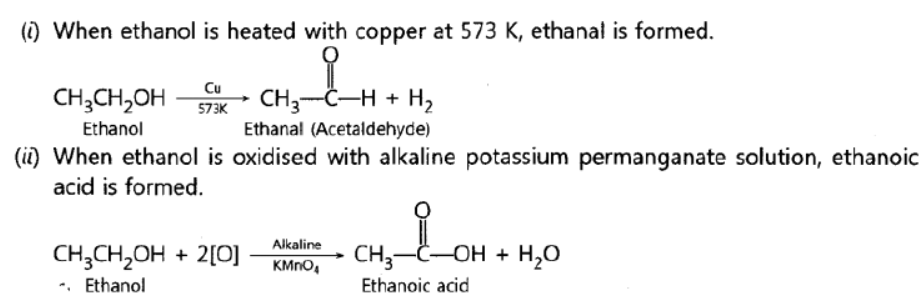
Question. Describe two examples of different oxidations of ethanol. Name the products obtained in each case.
Answer:
Question. Write a chemical equation in each case to represent the following types of chemical reactions of organic compounds:
(i) Oxidation reactions
(ii) Addition reactions
(iii) Substitution reactions
Answer:
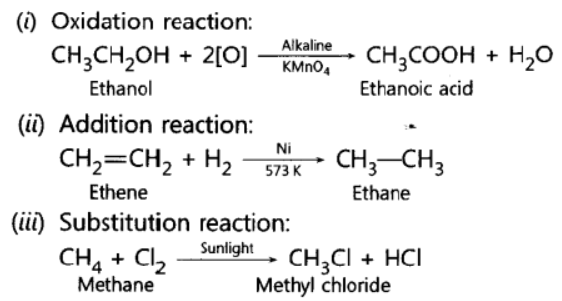
Question. Name the oxidising agent used for the conversion of ethanol to ethanoic acid.
Distinguish between ethanol and ethanoic acid on the basis of (i) litmus test, (ii) reaction with sodium hydrogencarbonate.
Answer: Alkaline potassium permanganate or Acidified potassium dichromate.
(i)Ethanol will not affect litmus paper. Ethanoic acid will turn blue litmus ‘ paper red.
(ii) Ethanol will not react with sodium hydrogen carbonate. Ethanoic acid will give brisk effervescence due to colourless, odourless carbon dioxide gas.
Question. Distinguish between esterification and saponification reactions of organic compounds with the help of the chemical equation for each. What is the use of (i) esters and (ii) saponification process?
Answer:

Question. Out of HCI and CH3COOH, which one is a weak acid and why? Describe an activity to support your answer.
Answer: Acetic acid ( CH3COOH) is a weaker acid because it does not dissociate completely into its ions in aqueous solution. .
Activity: Add zinc metal in HCI and CH3COOH respectively. The hydrogen gas will be evolved faster in HCI and slowly in CH3COOH. It shows acetic acid is a weak acid.
Alternative Method:
If we use pH paper, the colour of pH paper will be dark red in HCI and light red in CH3COOH which shows HCI is a strong acid and CH3COOH is a weak acid.
Question. a. Write chemical name and formula of vinegar?
b. Describe with a chemical equation what happens when sodium reacts with ethanol.
Answer :
a. Vinegar contains ethanoic acid,
b. Sodium ethoxide and hydrogen gas is formed.
2C2H5OH + 2Na → 2C2H5ONa + H2
Question. What is homologous series? Which of the following organic compounds belong to the same homologous series?
C2H6, C2H6O, C2H6O2, CH4O
Answer : The series of organic compounds having same functional group and similar chemical properties is called homologous series.
C2H6O(C2H5OH) and CH4O(CH3OH) belong to same homologous series.
Question. With the help of a suitable example explain in brief the process of hydrogenation mentioning the conditions of the reaction and also state any one physical property of substances changes due to hydrogenation.
Answer :
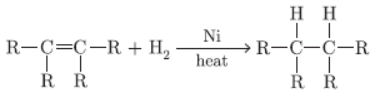
Liquid oil changes to solid ghee
Question. State reasons to explain why covalent compounds:
a. are bad conductors of electricity?
b. have low melting and boiling points?
Answer :
a. Covalent compounds do not form ions, hence they are bad conductor of electricity.
b. Covalent compounds have weak intermolecular forces of attraction, therefore, have low melting and boiling points.
Question. What is homologous series? Write the name and draw the structure of the second member of alkene series.
Answer :
a. The series of organic compounds having same functional group and similar chemical properties is called homologous series.
b.
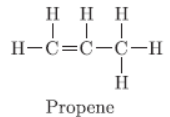
Question. List two characteristics of covalent compounds.
Answer :
(i) They have low melting and boiling point.
(ii) They do not conduct electricity.
Question. Write a chemical test to distinguish between ethanol and ethanoic acid.
or
How would you distinguish experimentally between ethanol and ethanoic acid with the help of sodium hydrogen carbonate? Write the chemical equation for the reaction involved.
Answer :
Add NaHCO3 to each of them separately. Ethanol will not react. Ethanoic acid will give brisk effervescence due to CO2.
CH3COOH + NaHCO3 → CH3COONa + H2O + CO2
C2H5OH + NaHCO3 → No reaction
Question. Explain the action of soap in removing an oily spot from a piece of cloth.
Answer :
Cleansing action of soap: Soap has ionic end which is hydrophilic, interacts with water while carbon chain is hydrophobic interacts with oil, grease. The soap molecules orient themselves in a cluster in which hydrophobic tails are inside the cluster and ionic ends face outside.
These cluster are called micelles. These attract oil which is washed away by water.
Question. Name an element, other than carbon, which exhibits property of catenation up to seven or eight atoms. Are these compounds stable?
Answer : Si and Sulphur (S8).
No, these compounds are not stable, rather they are reactive.
Question. Select alkenes and alkynes from the following:
C2H4, C3H4, C2H2, C4H8
Answer : Alkenes C2H4, C4H3 Alkynes C3H4, C2H2
Question. Why are detergents preferred over soaps for washing clothes in hard water? Explain.
Answer :
Detergents work well even with hard water because their calcium and magnesium salts are soluble in water. They do not form scum.
Question. What happens when ethyl alcohol and acetic acid react with each other in presence of cone. H2SO4?
Answer : Pleasant fruity smelling compound ester is formed cone. H2SO4.

Question. Name the functional groups of the following compounds:
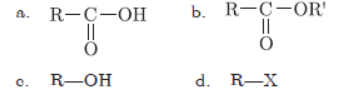
Answer :
a. Carboxylic acid
b. Ester
c. Alcohol
d. Halogen
Question. Why is hydrogenation? What is its industrial application?
Answer :
Hydrogenation is a process of adding hydrogen to unsaturated compounds in presence of catalyst like nickel to form saturated hydrocarbons. Industrially,
it is used to convert vegetable oils to vegetable ghee.
Question. List four characteristics of homologous series.
Answer :
a. All members are derived from same general formula.
b. All members have same functional group.
c. Each successive member differ by —CH2 unit.
d. All members can be prepared by same methods of preparation.
Question. Carbon does not form ionic compounds, why?
Answer :
Carbon cannot lose four electrons because high energy is needed to remove four electrons. It cannot gain 4 electrons because 6 protons cannot hold 10 electrons.
That is why carbon cannot form ionic compounds.
Question. What are soaps? Why do they form scum with hard water?
Answer :
Soaps are sodium or potassium salts of fatty acids e.g. sodium stearate. They react with Ca2+ and Mg2+ ions in hard water to form calcium and magnesium salt of fatty acids which are insoluble in water and called scum.
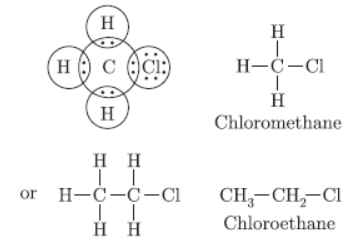
Question. Give the electron dot structure of chloro-methane. Also write the formula and the name of next homologue of it.
Answer :
Question. The structural formula of an ester is
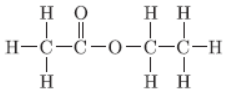
Write the structural formula of the corresponding alcohol and acid.
Answer :
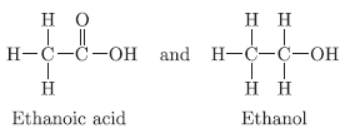
Question. How do the melting and boiling points of the hydrocarbons change with increase in molecular mass?
Answer :
Melting and boiling point of the hydrocarbons increases with increase in molecular mass because surface area increases which results an increase in vander Waal’s forces of attraction between molecules.
Question. Explain why cannot we have isomers of first three members of alkane family.
Answer : It is because branching is not possible with carbon atoms, that is why, there are no isomers till propane.
Question. Write balanced equations for the burning of (a) methane (b) ethane in air.
Answer : (a) CH4(g) + 2O2(g) $ CO2(g) + 2H2O(l)
(b) 2C2H6(g) + 7O2(g) $ 4CO2(g) + 6H2O(l)
Question. Draw electron dot structures of (i) C2H4 (ii) C2H5OH.
Answer :
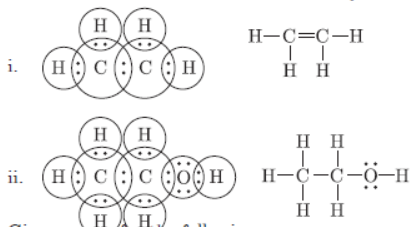
Question. Give reasons for the following:
a. Unsaturated hydrocarbons show addition reactions but not saturated hydrocarbons.
b. Carbon forms only covalent bonds.
Answer : a. Unsaturated hydrocarbons have double or triple bonds to which a molecule can be added whereas saturated hydrocarbons have single bonds addition reaction cannot take place.
b. It cannot lose four electrons because high energy is required. It cannot gain four electrons because 6 protons cannot hold 10 electrons. Thus it shares
four electrons to form covalent bonds and become stable.
Question. Write the name and molecular formula of an organic compound having its name suffixed with ‘—ol’ and having two carbon atoms in the molecule. With the help of a balanced chemical equation indicate what happens when it is heated with excess of cone. H2SO4.
Answer :(Img 7)
Question. Draw the structural formulae of the possible isomers for the compound with molecular formula C3H6O.
Answer :
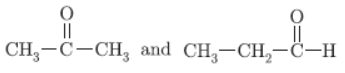
Question. Compare the structures of benzene and cyclohexane by drawing them.
Answer :
Benzene has 3 double bonds whereas cyclohexane has all single bonds.
Question. Write the names and molecular formulae of two organic compounds having functional group suffixed as ‘—oic acid’. With the help of a balanced chemical equation explain what happens when any one of them reacts with sodium hydroxide.
Answer :
HCOOH and CH3COOH
Methanoic acid Ethanoic acid
HCOOH + NaOH → HCOONa + H2O
It is an acid-base reaction where a salt i.e., sodium methanoate and H2O is formed.
Question. Draw the structures for the following compounds.
(i) Ethanoic acid
(ii) Bromopentane*
(iii) Butanone
(iv) Hexanal
Are structural isomers possible for bromopentane?
Answer.

(ii) There are many structural isomers possible for bromopentane. Among them, the structures of three isomers are given.

Question. Explain the nature of the covalent bond using the bond formation in CH3Cl.
Answer. Carbon can neither lose four of its electrons nor gain four electrons as both the processes require extra amount of energy and would make the system unstable. Therefore, it completes its octet by sharing its four electrons with other carbon atoms or with atoms of other elements. The bonds that are formed by sharing electrons are known as covalent bonds. In covalent bonding, both the atoms share the valence electrons, i.e., the shared electrons belong to the valence shells of both the atoms.
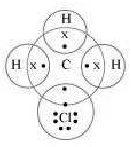
Here, carbon requires 4 electrons to complete its octet, while each hydrogen atom requires one electron to complete its duplet. Also, chlorine requires an electron to complete the octet. Therefore, all of these share the electrons and as a result, carbon forms 3 bonds with hydrogen and one with chlorine.
Question. Draw the electron dot structures for
(a) ethanoic acid.
(b) H2S.
(c) propanone.
(d) F2.
Answer.

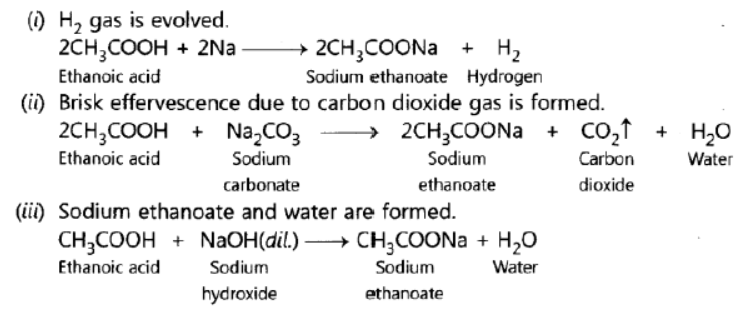
Question. Write chemical equations for what happens when
(i) sodium metal is added to ethanoic acid.
(ii) solid sodium carbonate is added to ethanoic acid.
(iii) ethanoic acid reacts with a dilute solution of sodium hydroxide.
Answer: