Please refer to Assignments Class 10 Science Acids Bases Salts Chapter 2 with solved questions and answers. We have provided Class 10 Science Assignments for all chapters on our website. These problems and solutions for Chapter 2 Acids Bases Salts Class 10 Science have been prepared as per the latest syllabus and books issued for the current academic year. Learn these solved important questions to get more marks in your class tests and examinations.
Acids Bases Salts Assignments Class 10 Science
Very Short Answer Questions
Question. What effect does an increase in concentration of H+(aq) ions in a solution have on pH of solution?
Answer : Increase in H+ concentration will lead to decrease in pH,
Question. Name a gas evolved when dilute HCl reacts with sodium hydrogen carbonate. How is it recognised?
Answer : Carbon dioxide, it turns lime water milky. In this way, CO2 gas is recognised.
Question. What would be the colour of litmus in a solution of sodium carbonate?
Answer : The red litmus will turn blue in Na2CO3 solution.
Question. What is the colour of litmus in a solution of ammonium hydroxide?
Answer : Red litmus will turn blue green colour in a solution of ammonium hydroxide.
Question. What is the difference between slaked lime and lime water?
Answer : The solid Ca(OH)2 is slaked lime whereas clear solution of Ca(OH)2 in water is lime water.
Question. Write a balanced chemical equation for the neutralisation reaction, mentioning the physical state of reactants and products.
Answer : NaOH(aq) + HCl(aq) → NaCl(aq) + H2O(l)
Question. On adding dilute hydrochloric acid to copper oxide powder, the solution formed is blue green. Predict the new compound formed which imparts a blue green colour to the solution.
Answer : Copper chloride imparts blue green colour to the solution.
Question. What are olfactory indicators?
Answer : Those indicators whose smell changes in acidic and basic solutions.
Question. Why does 1 M HCl solution have a high concentration of H+ ions than 1 M CH3COOH solution?
Answer :It is because 1M HCl is a strong acid and it is completely ionised in aqueous solution whereas CH3COOH is a weak acid, so it is only partially ionised.
Question. Write the chemical equation representing the action of atmospheric CO2 gas on bleaching powder when left exposed in open.
Answer : CaOCl2 + CO2 → CaCO3 + Cl2
Question. How will you test for the gas which is liberated when hydrochloric acid reacts with an active metal?
Answer : Bring a burning matchstick near the gas. If it is burnt with ‘pop’ sound, the gas is H2.
Question. How is the pH of solution of an acid influenced when it is diluted?
Answer : pH of the solution increases when it is diluted.
Question. At what pH rain water is said to be acidic ?
Answer : When pH < 5.5, the rain water becomes acidic.
Question. Which gas is evolved when dilute hydrochloric acid reacts with zinc metal? Write the molecular formula of this gas.
Answer : Dihydrogen gas, H2.
Question. Dry HCl gas does not change the colour of dry blue litmus. Give reason to justify it.
Answer : Dry HCl (g) does not form ions, therefore it does not affect dry blue litmus.
Question. Why is HCl a stronger acid than acetic acid?
Answer : HCl is completely ionised in aqueous solution whereas acetic acid is only partially ionised in aqueous solution.
Question. Arrange the following in an increasing order of their pH values: NaOH solution, Blood, Lemon juice.
Answer : Lemon juice < Blood < NaOH solution.
Question. At what pH in the mouth is tooth decay faster and why?
Answer : At pH lower than 5.5, tooth decay becomes faster because calcium phosphate (enamel) reacts with acid and gets corroded.
Question. Name the chemicals used in acid fire extinguisher and the gas evolved from it when it is used?
Answer : NaHCO3 (Sodium hydrogen carbonate) and H2SO4 (Sulphuric acid). The gas evolved is carbon dioxide.
Question. Which is a stronger acid, with pH = 5 or with pH=2?
Answer : The acid with pH = 2 is a stronger acid.
Question. A compound which is prepared from gypsum has the property of hardening when mixed with a proper quantity of water. Identify the compound and write its chemical formula.
Answer : CaSO4. 21H2O (Plaster of Paris), Calcium sulphate hemihydrate.
Question. What is meant by term pH of solution? The pH of rain water collected from two cities A’ and ‘B’ were found to be 6.0 and 5.0 respectively. The water of which city will be more acidic?
Answer : pH of solution is defined as negative logarithm of H+ion concentration. It determines the strength of acid and base. Rainwater with pH = 5 is more acidic.
Question. A few drops of sulphuric acid are added to water before electrolysis, why?
Answer : Water is not a good conductor of electricity. Few drops of sulphuric acid makes it better conductor of electricity.
Question. Write the names of two salts belonging to sodium family.
Answer : NaCl, Na2CO3 are two salts belonging to sodium family.
Short Answer Questions
Question. A compound which is prepared from gypsum has the property of hardening when mixed with a proper quantity of water. Identify the compound. Write the chemical equation for its preparation. For what purpose is it used in hospitals?
Answer: Plaster of Paris (CaSO4.1/2H2O) has the property of hardening when mixed with a proper quantity of water.
Plaster of Paris is obtained by heating gypsum at 373 K.
CaSO4.2H2O(s) 373 K → heat CaSO4.1/4 H2O+3/2H2O
Gypsum Plaster of Paris
Question. Write the chemical formula for washing soda. How may it be obtained from baking soda?
Name an industrial use of washing soda other than washing clothes.
Answer: The chemical formula of washing soda is Na2CO3.10H2O (sodium carbonate decahydrate).
It can be obtained by heating baking soda followed by recrystallisation from its aqueous solution.
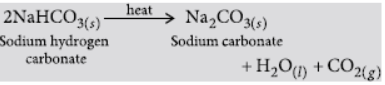
2NaHCO3 Heat → Na2CO3 + CO2 + H2O
Na2CO3 + 10H2O → Na2CO3.10H2O
The most important use of washing soda is in the manufacturing of glass.
Question. (i) Name the products formed when sodium hydrogen carbonate is heated.
(ii) Write the chemical equation for the reaction involved in the above.
In hospitals, it is used as plaster for setting the fractured bones in the right position.
Answer: (i) On heating sodium hydrogen carbonate (NaHCO3), it decomposes to give sodium carbonate and carbon dioxide.
Question. What effect does an increase in concentration of H+ (aq) in a solution have on the pH of solution?
Answer: As the concentration of H+ (aq) ions increases, the pH of the solution decreases.
Question. What is meant by ‘water of crystallisation’ of a substance? Describe an activity to show that blue copper sulphate crystals contain water of crystallisation.
Answer: Crystals of some salts contain certain amount of associated water. The water associated with the crystal (or molecule) of any salt is called water of crystallisation. e.g., CuSO4.5H2O, FeSO4.7H2O, etc.
Aim : To test the presence of water of crystallisation in copper sulphate crystals following activity is performed.
Materials required : Copper sulphate crystals, test tube holder, boiling tube, water, burner.
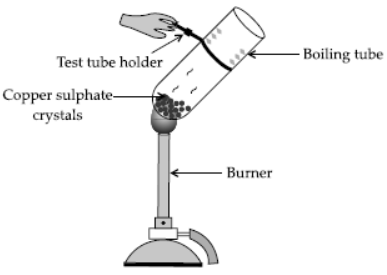
Procedure :
1. Take a few crystals of copper sulphate in a dry boiling tube. These have blue colour.
2. Heat the boiling tube by holding it with a test tube holder on the flaame of the burner.
Observations : You will observe that the colour of copper sulphate after heating becomes white. You may also notice water droplets in the boiling tube which are obtained from water of crystallisation.
After adding 2-3 drops of water on the white sample of copper sulphate (obtained after heating) you will observe that the blue colour of copper sulphate is restored.
CuSO4.5H2O Heat → CuSO4 + 5H2O
(Blue) (White)
Question. Fresh milk has a pH of 6. When it changes into curd (yogurt), will its pH value increase or decrease? Why?
Answer: Fresh milk has pH value equal to 6 but when it changes into curd (yogurt) then there is a slight decrease in pH value as curd contains lactic acid which is more acidic than milk. More acidic is a substance, lesser will be its pH.
Question. Write the chemical formula for bleaching powder. How is bleaching powder prepared?
For what purpose is it used in paper factories?
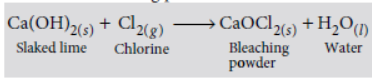
Answer: Chemical formula for bleaching powder is CaOCl2 (calcium oxychloride). It is prepared by reaction of slaked lime Ca(OH)2 with dry chlorine gas.
Ca(OH)2 + Cl2 → CaOCl2 + H2O
Bleaching powder is used for bleaching the wood pulp in paper factories.
Question. What happens when chlorine is passed over slaked lime at 313 K? Write chemical equation of the reaction involved and state two uses of the product obtained.
Answer: When chlorine is passed over slaked lime at 313 K then bleaching powder is formed.
Bleaching powder is used :
(i) for bleaching cotton fabres/fabrics in textile industries and wood pulp in paper industries.
(ii) as disinfectant and germicide.
Question. What is the chemical formula for Plaster of Paris? How is it prepared? State the common and chemical names of the compound formed when Plaster of Paris is mixed with water.
Answer: The chemical formula for Plaster of Paris is CaSO4.1/2H2O (calcium sulphate hemihydrate).
It is prepared by heating gypsum at 373 K.
CaSO4.2H2O 373 K → CaSO4.1/2 H2O + 3/2 H2O
Gypsum Plaster of Paris (POP)
When Plaster of Paris is mixed with water the compound formed is gypsum (common name).
The chemical name of gypsum is calcium sulphate dihydrate.
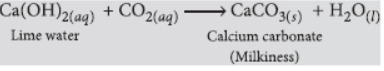
Question. Name the gas evolved when dilute HCl reacts with sodium hydrogen carbonate. How is it recognised?
Answer: When sodium hydrogen carbonate reacts with dilute HCl then carbon dioxide (CO2) gas is released.
NaHCO3(s) + HCl(aq) → NaCl(aq) + H2O(l) + CO2(g)
This gas is recognised by passing it through lime water which turns milky due to the formation of calcium carbonate.
Question. A white chemical compound becomes hard on mixing proper quantity of water. It is also used to maintain joints in fixed position. Name the chemical compound and write its chemical formula. Write the chemical equation to show what happens when water is added to this compound in proper quantity.
Answer : CaSO4. 1/2 H2O is the formula of the compound. The name of compound is ‘Plaster of Paris’ (Calcium sulphate hemihydrate).
CaSO4 .1/2H2O + 3/2H2O CaSO4 . 2H2O
(Plaster of Paris) (Gypsum)
Question. Two solutions ‘A’ and ‘B’ have pH value 3.0 and 10.5 respectively. Which of these will turn
a. Blue litmus solution to red,
b. Phenolphthalein from colourless to pink? Justify your answer in each case.
Answer : a. ‘A’ with pH = 3, will turn blue litmus red because it is acidic in nature.
b. “B’ with pH = 10.5, will turn phenolphthalein colourless to pink because ‘B’ is basic in nature.
Question. The pH of soil ‘A’ is 7.5, while that of soil “B is 4.5.
Which of the two soils A or B should be treated with powdered chalk to adjust the pH and why?
Answer : Soil ‘B’ is acidic, therefore it needs to be treated with powdered chalk to adjust its pH because chalk is basic, which will make soil neutral.
Question. Write the chemical equation to describe how baking soda is produced on a large scale. Also write the chemical name of the products formed in the reaction.
Answer :
NH3 + H2O + CO2 + NaCl → NaHCO3 + NH4Cl
Sodium hydrogen Ammonium
carbonate chloride
Question. What is chlor-alkali process? Write a balanced chemical equation for the reaction involved in this process, to justify your answer.
Answer :
When brine solution is electrolysed we get alkali (NaOH) and chlorine (Cl2) gas, this process is called chlor-alkali process.
2NaCl (aq) + 2H2O(l) → 2NaOH(aq) + H2(g) + Cl2(g)
Question. What is the action of litmus on
a. dry ammonia gas
b. solution of ammonia gas in water?
Answer : a. There is no effect of dry litmus on dry ammonia gas.
b. Solution of ammonia will turn red litmus blue.
Question. State the observations you would make on adding sodium hydroxide to an aqueous solution of
a. ferrous sulphate,
b. aluminium chloride.
Answer : a. Green precipitate of Fe(OH)2 will be formed:
FeSO4 (aq) + 2NaOH(aq) → Fe(OH) ↓ NaSO4 (aq)
(Green ppt)
b. White precipitate of Al(OH)3 will be formed:
AlCl (aq) + 3NaOH → Al(OH)4 (s) + 3NaCl (aq)
(White ppt)
Question. The pH of the mouth of a person is lower I than 5.5.
What changes will occur in his mouth? How these changes can be controlled? Write any two measures.
Answer : Acid will be formed in the mouth which causes tooth decay.
a. Wash your mouth with water after every meal.
b. Brush your teeth after meal. Toothpastes are basic in nature and it will neutralise the acid formed in mouth.
Question. What is a neutralisation reaction? Give one example.
Answer :
The reaction in which acid reacts with a base to form salt and water is called neutralisation reaction e.g.,
KOH(aq) + HNO3(aq) → KNO3(aq) + H2O(l)
Question. 15 mL of water and 10 mL of sulphuric acid are to be mixed in a beaker
a. State the method that should be followed with reason.
b. What is this process called?
Answer :
a. Acid should be added to the water slowly with constant cooling because the reaction is highly exothermic.
b. This process is called dilution.
Question. Name the acid present in the following:
a. Tomato,
b. Vinegar,
c. Tamarind
Answer :
a. Tomato contains oxalic acid.
b. Vinegar contains acetic acid.
c. Tamarind contains tartaric acid.
Question. a. Define pH scale. Draw a figure showing variation of pH with change in concentration of H+(aq) and OH-(aq) ions.
b. Mention the pH of acidic, basic and neutral solutions respectively.
Answer :
a. pH scale is a scale which is used for measuring hydrogen ion concentration in a solution. (Img 8)
b. pH < 7 is for acidic solution, pH > 7 basic solution,
pH = 7 for neutral solution.
Question. a. Define olfactory indicators. Name two substances which can be used as olfactoiy indicators.
b. Choose strong acids from the following:
CH3COOH, H2SO4, H2CO3, HNO3
Answer :
a. Olfactory indicators: They give different smell in acidic and basic medium e.g., onion, clove, vanilla.
b. HNO3 and H2SO4 are strong acids among the given acids.
Question. Explain the action of dilute hydrochloric acid on the following with suitable chemical equations:
a. Magnesium ribbon,
b. Sodium hydroxide,
c. Crushed egg shells.
Answer :
a. Mg(s) + 2HCl(dil) → MgCl2(aq) + H2(g)
b. NaOH(aq) + HCl(dil) → NaCl(aq) + H2O(l)
c. CaCO3 + 2HCl dil →CaCl2 g + CO2 g + H2O
Question. You are provided with magnesium ribbon and sulphur powder. Explain with the help of activity that metal oxides are basic and oxides of non-metals are acidic in nature.
Answer :
Bum magnesium ribbon with the help of tongs to form white ash. Dissolve the ash in hot water. Add red litmus which turns blue, showing that MgO is a basic oxide.
2Mg(s) + O2(g) → 2MgO(s)
MgO(s) + H2O(Hot) → Mg(OH)2(aq)
Heat sulphur taken in a iron spatula and pass the gas through water. Add blue litmus into it. It will turn red showing SO2 is an acidic oxide.
S + O2 → SO2
SO2 + H2O → H2SO3 (Sulphurous acid)
Question. List two differences between acids and bases on the basis of chemical properties.
Answer : (Table 8)
Question. A substance ‘X’ is used as antacid reacts with hydrochloric acid to produce a gas W which is used in fire extinguishers:
a. Name the substance X and ‘Y’.
b. Write a balanced equation of the reaction between X and hydrochloric acid.
Answer :
a. ‘X’ is NaHCO3 (Sodium hydrogen carbonate). ‘Y’ is CO2 gas, which is used in fire extinguishers.
b. NaHCO3(s) + HCl (aq) → NaCl (aq) + H2O(l) + CO2(g)
Question. “Sodium hydrogen carbonate is a basic salt.” Justify the statement. How is it converted into washing soda? Explain.
Answer :
NaHCO3 is a salt of NaOH which is a strong base and H2CO3 (Carbonic acid) which is a weak acid, therefore it is a basic salt. It can be converted into washing soda by heating followed by crystallisation:
2NaHCO (s) → Heat Na2CO3 (s) + CO2 (g) + H2O(l)
Na2CO3 + 10H2O → Na2CO3 . 10H2O
(washing soda)
Question. Explain how antacid works.
Answer : Antacids are weakly basic in nature. They neutralise excess of HCl present in our stomach and gives us relief from hyper-acidity.
Question. Equal lengths of magnesium ribbon are taken in test tube ‘A’ and ‘B’. Hydrochloric acid (HCl) is added to test tube A’ while acetic acid (CH3COOH) is added to test tube ‘B’. In which test tube, will fizzing occur more vigorously and why?
Answer : The fizzing will occur more vigorously in test tube ‘A’ because HCl is a strong acid and reacts faster than acetic acid which is a weak acid.
Question. State what does pH of solution signify? Three solutions A, B and C have pH values of 6, 2 and 10 respectively.
Which one of these solutions is highly acidic? Which solution will turn red litmus blue?
Answer : pH of solution signifies the nature of the solution i.e.,
it is weakly acidic, strongly 1 acidic, neutral, weakly basic, strongly basic.
‘B’ with pH = 2 is strongly acidic.
‘C’ with pH = 10 will turn red litmus blue.
Question. Define an acid and a base. Name one weak acid and one strong acid.
Answer : Acid is a substance which gives H+ ions in an aqueous solution.
Base is substance which gives OH- ions in the aqueous solution.
CH3COOH is a weak acid, H2SO4 is a strong acid.
Question. What is universal indicator? State the purpose for which this indicator is used.
Answer : Universal indicator is a mixture of a number of indicators. It is used to determine pH of a solution.
Question. Name the natural source of each of the following acid:
a. Citric acid,
b. Oxalic acid,
c. Lactic acid,
d. Tartaric acid.
Answer :
a. Citric acid—Lemon, Orange.
b. Oxalic acid—Tomato, Guava
c. Lactic acid—Curd, Sour milk
d. Tartaric acid—Tamarind
Question. Explain why sodium hydroxide solution cannot be kept in aluminium containers? Write the equation for the reaction that may take place for the same.
Answer : It because ‘Al’ reacts with NaOH to form sodium meta-aluminate and hydrogen gas:
2Al + 2NaOH + 2H2O → NaAlO2+3H2
Question. A student detected the pH of four unknown solutions
A, B, C and D as follows: 11, 5, 7 and 2. Predict the nature of these solutions.
Answer :
pH = 11 is basic
pH = 5 is acidic
pH = 7 is neutral
pH = 2 is strongly acidic
Question. What is meant by the term water of crystallisation?
How would you show that copper sulphate crystals contains water of crystallisation?
Answer : The molecules of water associated with a crystalline substance are called water of crystallisation.
When hydrated copper sulphate is heated its colour changes from blue to dirty white and water droplets are formed.
CuSO4.5H2O →Heat CuSO4 + 5H2O
If we add little water to anhydrous CuSO4, we get blue colour again. It is the presence of molecules of water of crystallisation which was lost on heating.
CuSO4 + 5H2O → CuSO4.5H2O
(Anhydrous)
Question. Mention the pH of aqueous solution of the following salts as 7, more than 7, less than 7.
KCl, Na2CO3, NH4C1, NaNO3 (Sodium nitrate)
Answer :
KCl and NaNO3 has pH = 7
Na2CO3 has pH > 7
NH4Cl has pH < 7
Question. Give two uses of baking soda and washing soda each.
Answer : Use of baking soda:
a. It is used in making of bread, biscuits, cakes.
b. It is used as an antacid.
Use of washing soda:
a. It is used as a cleansing agent.
b. It is used to remove hardness of water.
Question. A compound ‘X’ of sodium is commonly used for making crispy pakoras. It is also used for curing acidity in the stomach. Identify ‘X’. Write the formula and its chemical name. State the reaction which takes place when it is heated.
Answer :
‘X’ is NaHCO3, sodium hydrogen carbonate. It is used in cooking and for curing acidity in stomach.
2NaHCO3 Heat Na2CO3 + CO2 +H2O
Long Answer Questions
Question. a. The blue colour of crystals of a substance on heating in a closed test tube gets changed but the colour was regained after sometime on cooling.
Name that substance and write its chemical formula. Explain the phenomenon involved.
b. Write name and chemical formulae of two such compounds whose one unit is associated with 10 and 2 water molecules respectively.
Answer :
a. Hydrated copper sulphate, CuSO4.5H2O is the name and chemical formula of that substance.
It loses water of crystallisation on heating and regains these molecules of water on exposure to the atmosphere:
CuSO . 5H2O → HeatCuSO + 5H2O
CuSO4 (s) + 5H2O(l) →CuSO4 . 5H2O
Blue
b. Na2CO3.10H2O, washing soda (Sodium carbonate decahydrate) has 10 molecules of water of crystallisation. CaSO4.2H2O, gypsum, chemically calcium sulphate dihydrate has 2 molecules of water of crystallisation.
Question. Describe an activity with diagram to illustrate that the reaction of metal carbonates or metal bicarbonates with acid produces carbon dioxide. Write the relevant equations of all the reactions that take place. Name any two forms in which calcium carbonate is found in nature.
Answer :
Activity: To show reaction of metal carbonates and metal hydrogen carbonates with dilute acids.
1. Take marble chips in Woulfe bottle.
2. Set the apparatus as shown in the diagram.
3. Add dilute HCl with the help of thistle funnel.
4. Collect the gas and pass through lime water and bring a burning matchstick near the gas.
5. Observe what happens.
Observation: Lime water turns milky. The burning matchstick gets extinguished.
Conclusion: Metal carbonates react with dilute acids to liberate carbon dioxide.
Repeat the experiment with NaHCO3 taken in Woulfe bottle.
Observation: CO2 gas will be evolved which turns lime water milky.
Conclusion: Metal hydrogen carbonates give CO2 with dilute acids. (Img 9)NaHCO3 + HCl → NaCl + H2O + CO2
Na2CO3 + 2HCl → 2NaCl + H2O + CO2
Question. What is neutralisation reaction? Give two examples.
Answer :
The reaction in which acid reacts with base to form salt and water is called neutralisation reaction e.g.,
NaOH + HCl → NaCl + H2O
2KOH + H2SO4 → K2SO4 + 2H2O
CaCO3 (Marble), CaCO3(Chalk) are the two forms in which calcium is found in nature.
Question. Five solutions A, B, C, D, and E showed pH as 4, 7,
1, 11 and 9 respectively when tested with universal indicator. Which solution is
a. Neutral,
b. Strongly alkaline,
c. Strongly acidic,
d. Weakly acidic,
e. Weakly alkaline.
Arrange the pH in increasing order of H+ ion concentration.
Answer :
a. ‘B’ is neutral,
b. D is strongly alkaline,
c. ‘C’ is strongly acidic,
d. A is weakly acidic,
e. ‘E’ is weakly basic. D<E<B<A<C is the increasing order of H+ ion concentration.
Question. You have been provided with three test tubes. One of them contains distilled water and the other two contains an acidic solution and a basic solution respectively. If you are given only red litmus, how will you identify the contents 5 of each test tube?
Answer : Add red litmus to each of them. The test tube in which it turns blue contains the base.
Add blue litmus to the remaining two test tubes.
The one in which it turns red contains the acid. The other one in which blue litmus and red litmus do not change contains distilled water,
108. While constructing a house, a builder selects marble flooring and marble table top for the kitchen where vinegar and lemon juice, tamarind etc., and more often used for cooking are to be kept. Will you agree
to this selection and why?
Answer : No, he has taken wrong decision. Marble will react with vinegar and other acids and get corroded.
CaCO3 + 2CH3COOH → (CH2COO)2Ca + H2O + CO2
Question. Name the products formed in each case when:
a. Hydrochloric acid reacts with caustic soda.
b. Granulated zinc reacts with caustic soda.
c. Carbon dioxide is passed through lime water.
Answer :
a. NaOH + HCl → NaCl + H2O
Sodium chloride Water
b. Zn + 2NaOH → Na2ZnO2 + H2 (g)
soudium zincate Hydrogen
c. Ca(OH)2 + CO2 (g) → CaCO3 (s) + H2O(l)
Calcium carbonate Water
Question. a. Define indicator. Name two indicators obtained from plants.
b. Write a balanced chemical equation for the reaction taking place when sodium oxide reacts with water. How will this solution behave towards phenolphthalein and red litmus paper?
c. State what happens when sodium hydroxide solution reacts with hydrochloric acid.
Answer :
a. Indicator is a substance which give different colour or odour in acid and base e.g., litmus and turmeric are indicators obtained from plants.
b. Na2O(s) + H2O(l) → 2NaOH(aq)
Solution will turn phenolphthalein pink and red litmus paper blue.
c. Sodium chloride and water are formed:
NaOH(aq) + HCl(aq) → NaCl(aq) + H2O(l)
Question. State the reason for the following statements:
a. Tap water conducts electricity whereas distilled water does not.
b. Dry hydrogen chloride gas does not turn blue litmus red whereas dilute hydrochloric acid does.
c. During summer season, a milkman usually adds a very small amount of baking soda to fresh milk.
d. For dilution of an acid, acid is added to water and not water to acid.
e. Ammonia is a base but it does not contain hydroxyl group.
Answer :
a. Tap water contains ions which makes it a good conductor whereas distilled water does not contain any ions.
b. Dry HCl gas does not dissociate into ions, so it has no effect on the litmus. Hydrochloric acid form ions, so it turns blue litmus red.
c. Baking soda prevents the formation of lactic acid when milk turns sour.
d. Acid is added to water slowly because the reaction is highly exothermic. If water is added to acid, then glass container may break due to lot of heat evolved.
e. NH3 dissolves in H2O forming NH4OH, therefore it acts as base:
NH3 + H2O → NH4OH → NH4 + OH-
Question. a. State the chemical properties on which the following uses of baking soda are based:
(i) as an antacid,
(ii) as a soda acid fire extinguisher,
(iii) to make bread and cake soft and spongy.
b. How is washing soda obtained from baking soda?
Write the relevant balanced chemical equation.
Answer :
a. (i) It is basic in nature.
(ii) It liberates CO2 with acid which extinguishes fire.
(iii) It releases CO2 gas on heating which makes bread and cake soft and spongy.
b. Washing soda is obtained by heating baking soda followed by crystallisation:
2NaHCO3(s) → Na2CO3 + CO2 + H2O
Na2CO3 + 10H2O → Na2CO . 10H O
(Washing soda)
Question. Write balanced chemical equations for the following:
a. Bleaching powder is kept open in air.
b. Blue crystals of copper sulphate are heated.
c. Chlorine gas is passed through dry slaked lime.
d. Carbon dioxide gas is passed through lime water.
e. NaOH solution is heated with zinc granules.
Answer :
a. CaOCl2 + CO2 → CaCO3 + Cl2
b. CuSO4.5H2O → Heat CuSO4 + 5H2O
c. Ca(OH)2 + Cl2 → CaOCl2 + H2O
d. Ca(OH)2 + CO2 → CaCO3 + H2O
e. Zn + 2NaOH → Na2ZnO2 + H2
Question. Equal length of magnesium ribbon are taken in two test tubes A and B. H2SO4 is added to test tube ‘A’ and H2CO3 is added in test tube ‘B’ in equal amounts:
a. Identify the test tube showing vigorous reaction.
b. Give reason to support your answer.
c. Name the gas liberated in both the test tubes. How will you prove its liberation?
d. Write chemical equations for both the reactions.
e. Out of two acids taken above, which one will have lower pH value and lower H+ ion concentration respectively?
Answer : a. ‘A’ will show vigorous reaction.
b. H2SO4 is a strong acid, it reacts faster than H2CO3,
a weak acid.
c. H2 gas. If we bring a burning splinter near the gas, it will burn with ‘pop’ sound.
d. Mg + H2SO4 → MgSO4 + H2
Mg + H2CO3 → MgCO3 + H2
e. H2SO4 will have lower pH. H2CO3 will have lower H+ ion concentration,


