Please refer to the Some Basic Concepts of Chemistry Revision Notes given below. These revision notes have been designed as per the latest NCERT, CBSE and KVS books issued for the current academic year. Students will be able to understand the entire chapter in your class 11th Chemistry book. We have provided chapter wise Notes for Class 11 Chemistry as per the latest examination pattern.
Revision Notes Chapter 1 Some Basic Concepts of Chemistry
Students of Class 11 Chemistry will be able to revise the entire chapter and also learn all important concepts based on the topic wise notes given below. Our best teachers for Grade 11 have prepared these to help you get better marks in upcoming examinations. These revision notes cover all important topics given in this chapter.
Chemistry: Chemistry is the branch of science that deals with the composition, structure and properties of matter. Chemistry is called the science of atoms and molecule
Branches of Chemistry
- Organic Chemistry – This branch deals with study of carbon compounds especially hydrocarbons and their derivatives. Inorganic Chemistry-This branch deals with the study of compounds of all other elements except carbon. It largely concerns itself with the study of minerals found in the Earth’s crust.
- Physical Chemistry- The explanation of fundamental principles governing various chemical phenomena is the main concern of this branch. It is basically concerned with laws and theories of the different branches of chemistry. Industrial Chemistry-The chemistry involved in industrial processes is studied under this branch.
- Analytical Chemistry- This branch deals with the qualitative and quantitative analysis of various substances. Biochemistry-This branch deals with the chemical changes going on in the bodies of living organisms; plants and animals.
- Nuclear Chemistry- Nuclear reactions, such as nuclear fission, nuclear fusion, transmutation processes etc. are studied under this branch.
PROPERTIES OF MATTER AND THEIR MEASUREMENT-Every substance has unique or characteristic properties. These properties can be classified into two categories – physical properties and chemical properties. - Physical properties- Physical properties are those properties which can be measured or observed without changing the identity or the composition of the substance. E.g. colour, odour, melting point, boiling point, density etc.
The measurement or observation of chemical properties requires a chemical change to occur. e.g. Burning of Mg-ribbon in air - Chemical properties- Chemical properties are characteristic reactions of different substances; these include acidity or basicity, combustibility etc.Many properties of matter such as length, area, volume, etc., are quantitative in nature.
Metric System was based on the decimal system.
The International System of Units (SI)
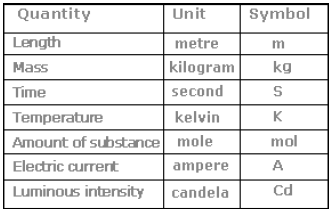
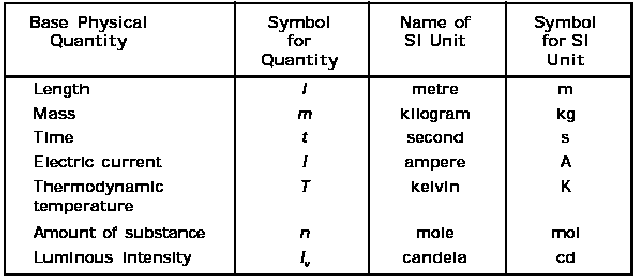
The International System of Units (in French Le Systeme International d’Unites– abbreviated as SI) was established by the 11th General Conference on Weights and Measures (CGPM from ConferenceGenerale des Poids at Measures). The SI system has seven base units
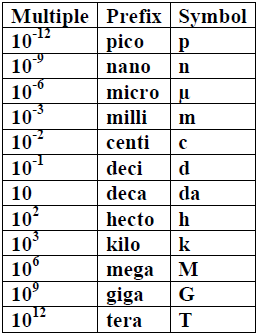
Prefixes in SI system
Mass and Weight– Mass of a substance is the amount of matter present in it while
weight is the force exerted by gravity on an object. The mass of a substance is constant whereas its weight may vary from one place to another due to change in gravity. The mass of a substance can be determined very accurately by using an analytical balance
Volume- Volume has the units of (length)3. So volume has units of m3 or cm3 or dm3.A common unit, litre (L) is not an SI unit, is used for measurement of volume of liquids. 1 L = 1000 mL, 1000 cm3 = 1 dm3
Density- Density of a substance is its amount of mass per unit volume.SI unit of density = SI unit of mass/SI unit of volume = kg/m3 or kg m–3This unit is quite large and a chemist often expresses density in g cm–3.
Temperature-There are three common scales to measure temperature — °C (degree celsius), °F (degree Fahrenheit) and K (kelvin). Here, K is the SI unit.
°F=9/5(°C)+32
K = °C + 273.15
Note—Temperature below 0 °C (i.e. negative values) are possible in Celsius scale but in Kelvin scale, negative temperature is not possible.
Scientific Notation
In which any number can be represented in the form N × 10n (Where n is an exponent having positive or negative values and N can vary between 1 to 10).
e.g. We can write 232.508 as 2.32508 x102 in scientific notation. Similarly, 0.00016 can be written as 1.6 x 10–4
Precision refers to the closeness of various measurements for the same quantity.
Accuracy is the agreement of a particular value to the true value of the result
Significant Figures
The reliability of a measurement is indicated by the number of digits used to represent it. To express it more accurately we express it with digits that are known with certainty. These are called as Significant figures. They contain all the certain digits plus one doubtful digit in a number.
Rules for Determining the Number of Significant Figures
- All non-zero digits are significant. For example, 6.9 has two significant figures, while 2.16 has three significantfigures. The decimal place does not determine the number of significant figures.
- A zero becomes significant in case it comes in between non zero numbers. For example, 2.003 has four significant figures, 4.02 has three significant figures.
- Zeros at the beginning of a number are not significant. For example, 0.002 has one significant figure while 0.0045 has two significant figures.
- All zeros placed to the right of a number are significant. For example, 16.0 has three significant figures, while 16.00 has four significant figures. Zeros at the end of a number without decimal point are ambiguous.
- In exponential notations, the numerical portion represents the number of significant figures. For example, 0.00045 is expressed as 4.5 x 10–4 in terms of scientific notations. The number of significant figures in this number is 2, while in Avogadro’s number (6.023 x 1023 ) it is four.
- The decimal point does not count towards the number of significant figures. For example, the number 345601 has six significant figures but can be written in different ways, as 345.601 or 0.345601 or 3.45601 all having same number of significant figures.
Retention of Significant Figures – Rounding off Figures
The rounding off procedure is applied to retain the required number of significant figures.
1. If the digit coming after the desired number of significant figures happens to be more than 5, the preceding significant figure is increased by one, 4.317 is rounded off to 4.32.
2. If the digit involved is less than 5, it is neglected and the preceding significant figure remains unchanged, 4.312 is rounded off to 4.31.
3. If the digit happens to be 5, the last mentioned or preceding significant figure is increased by one only in case it happens to be odd. In case of even figure, the preceding digit remains unchanged. 8.375 is rounded off to 8.38 while 8.365 is rounded off to 8.36.Dimensional Analysis During calculations generally there is a need to convert units from one system to other. This is called factor label method or unit factor method or dimensional analysis. For example- 5 feet and 2 inches (height of an Indian female) is to converted in SI unit
1 inch = 2.54 x 10–2 m
1=2.54 × 10–2 m/1 inch then, 5 feet and 2 inch = 62 inch
=62 inch × 2.54 × 10–2 m/1 inch=1.58 m
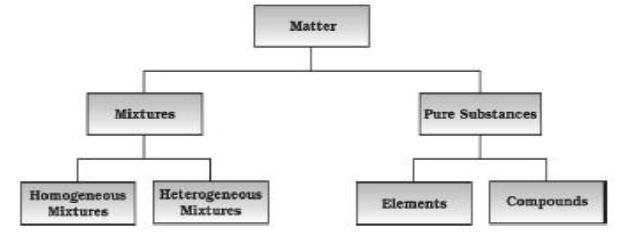
Physical Classification of Matter
Chemical Classification of matter-
Elements
An element is the simplest form of matter that cannot be split into simpler substances or built from simpler substances by any ordinary chemical or physical method. There are 114 elements known to us, out of which 92 are naturally occurring while the rest have been prepared artificially. Elements are further classified into metals, non-metals and metalloids
Compounds
A compound is a pure substance made up of two or more elements combined in a definite proportion by mass, which could be split by suitable chemical methods.
Characteristics of compound
- Compounds always contain a definite proportion of the same elements by mass.
- The properties of compounds are totally different from the elements from which they are formed.
- Compounds are homogeneous.
- Compounds are broadly classified into inorganic and organic compounds. Inorganic compounds are those, which are obtained from non-living sources such as minerals. For example, common salt, marble and limestone. Organic compounds are those, which occur in living sources such as plants and animals. They all contain carbon. Common organic compounds are oils, wax, fats etc.
Mixtures
A mixture is a combination of two or more elements or compounds in any proportion so that the components do not lose their identity. Air is an example of a mixture Mixtures are of two types, homogeneous and heterogeneous.
Homogeneous mixtures have the same composition throughout the sample. The components of such mixtures cannot be seen under a powerful microscope. They are also called solutions. Examples of homogeneous mixtures are air, seawater, gasoline, brass etc.
Heterogeneous mixtures consist of two or more parts (phases), which have different compositions. These mixtures have visible boundaries of separation between the different constituents and can be seen with the naked eye e.g., sand and salt, chalk powder in water etc.
LAWS OF CHEMICAL COMBINATIONS
Law of Conservation of Mass (Given by Antoine Lavoisier in 1789).
It states that matter (mass) can neither be created nor destroyed.
Law of Definite Proportions or Law of Constant Composition:
This law was proposed by Louis Proust in 1799, which states that:
‘A chemical compound always consists of the same elements combined together in the same ratio, irrespective of the method of preparation or the source from where it is taken’.
Law of Multiple Proportions Proposed by Dalton in 1803, this law states that:
‘When two elements combine to form two or more compounds, then the different masses of one element, which combine with a fixed mass of the other, bear a simple ratio to one another’.
Gay Lussac’s Law of Gaseous Volumes (Given by Gay Lussac in 1808.)
According to this law when gases combine or are produced in a chemical reaction they do so in a simple ratio by volume provided all gases are at same temperature and pressure.
e.g.H2(g) + Cl2(g) —→2HCl(g)
1V 1V 2V
All reactants and products have simple ratio 1:1:2.
Avogadro Law (In 1811, Given by Avogadro)
According to this law equal volumes of gases at the same temperature and pressure should contain equal number of molecules.
Dalton’s Atomic Theory
- All substances are made up of tiny, indivisible particles called atoms.
- Atoms of the same element are identical in shape, size, mass and other properties.
- Atoms of different elements are different in all respects.
- Atom is the smallest unit that takes part in chemical combinations.
- Atoms combine with each other in simple whole number ratios to form compound atoms called molecules.
- Atoms cannot be created, divided or destroyed during any chemical or physical change
Atoms and Molecules
The smallest particle of an element, which may or may not have independent existence is called an atom, while the smallest particle of a substance which is capable of independent existence is called a molecule.
Molecules are classified as homoatomic and heteroatomic. Homoatomic molecules are made up of the atoms of the same element and heteroatomic molecules are made up of the atoms of the different element have different atomicity (number of atoms in a molecule of an element) like monoatomic, diatomic, triatomic and polyatomic.
Atomic Mass Unit
One atomic mass unit is defined as a mass exactly equal to one twelfth the mass of one carbon -12 atom. And 1 amu = 1.66056×10–24 g.
Today, ‘amu’ has been replaced by ‘u’ which is known as unified mass.
Atomic Mass
Atomic mass of an element is defined as the average relative mass of an atom of an element as compared to the mass of an atom of carbon -12 taken as 12.
Atomic mass= mass of an atom / 1/12 mass of a carbon atom (12 C)
Gram Atomic Mass
The quantity of an element whose mass in grams is numerically equal to its atomic mass. In simple terms, atomic mass of an element expressed in grams is the gram atomic mass or gram atom.
For example, the atomic mass of oxygen = 16 amu
Therefore gram atomic mass of oxygen = 16 g
Molecular Mass
Molecular mass of a substance is defined as the average relative mass of its molecule as compared to the mass of an atom of C-12 taken as 12. It expresses as to how many times the molecule of a substance is heavier than 1/12th of the mass of an atom of carbon.
For example, a molecule of carbon dioxide is 44 times heavier than 1/12th of the mass of an atom of carbon. Therefore the molecular mass of CO2 is 44 amu.
It is obtained by adding the atomic masses of all the atoms present in one molecule.
Gram Molecular Mass
A quantity of substance whose mass in grams is numerically equal to its molecular mass is called gram molecular mass. In simple terms, molecular mass of a substance expressed in grams is called gram molecular mass.
e.g., the molecular mass of oxygen = 32 amu
Therefore, gram molecular mass of oxygen = 32 g
Formula Mass-
Sum of atomic masses of the elements present in one formula unit of a compound. It is used for the ionic compounds.
Mole Concept.
Mole is defined as the amount of a substance, which contains the same number of chemical units (atoms, molecules, ions or electrons) as there are atoms in exactly 12 grams of pure carbon-12.
A mole represents a collection of 6.022 x1023( Avogadro’s number) chemical units..
The mass of one mole of a substance in grams is called its molar mass.
Molar Volume
The volume occupied by one mole of any substance is called its molar volume. It is denoted by Vm. One mole of all gaseous substances at 273 K and 1 atm pressure occupies a volume equal to 22.4 litre or 22,400 mL. The unit of molar volume is litre per mol or millilitre per mol
PERCENTAGE COMPOSITION
The mass percentage of each constituent element present in any compound is called its percentage composition
Mass % of the element= Mass of element in 1 molecule of the compound/Molecular mass of the compound x 100
Empirical Formula and Molecular Formula
An empirical formula represents the simplest whole number ratio of various atoms present in a compound. E.g. CH is the empirical formula of benzene.
The molecular formula shows the exact number of different types of atoms present in a molecule of a compound. E.g. C6H6 is the molecular formula of benzene.
Relationship between empirical and molecular formulae
The two formulas are related as Molecular formula = n x empirical formula
n= Molecular mass / empirical formula mass
Chemical Equation-
Shorthand representation of a chemical change in terms ofsymbols and formulae of the substances involved in the reaction is called chemical equation..
The substances that react among themselves to bring about the chemical changes are known as reactants, whereas the substances that are produced as a result of the chemical change, are known as products
Limiting Reagent- The reactant which gets consumed first or limits the amount of product formed is known as limiting reagent
Reactions in Solutions- The concentration of a solution can be expressed in any of the following ways
1. Mass Percent is the mass of the solute in grams per 100 grams of the solution.
mass % of the solute= mass of the solute / mass of the Solution x 100
A 5 % solution of sodium chloride means that 5 g of NaCl is present in 100g of the solution.
2. Volume percent is the number of units of volume of the solute per 100 units of the volume of solution.
volume % of the solute = volume of the solute / volume of the solution x 100
A 5 % (v/v) solution of ethyl alcohol contains 5 cm3 of alcohol in 100 cm3 of the solution
3. Molarity of the solution is defined as the number of moles of solute dissolved per litre (dm3) of the solution. It isdenoted by the symbol M. Measurements in Molarity can change with the change in temperature because solutions expand or contract accordingly.
Molarity of the solution = No. of moles of the solute / Volume of the solution in litre = n/V
The Molarity of the solution can also be expressed in terms of mass and molar mass
Molarity of the solution = Mass of the solute / Molar mass of the solute x volume of the solution in liter
In terms of weight, molarity of the substance can be expressed as:
molarity =Wg/ Mg mol-1 x V litre =W/Mx V mol/L
Molarity equation
To calculate the volume of a definite solution required to prepare solution of other molarity, the following equation is used:
M1V1 = M2V2, where M1= initial molarity, M2= molarity of the new solution, V1= initial volume and V2= volume of the new solution.
4. Molality- Molality is defined as the number of moles of solute dissolved per 1000 g (1 kg) of solvent. Molality is expressed as ‘m’.
Molality = Moles of the solute / Wt. of solvent (in gm) x 1000
5. Mole Fraction is the ratio of number of moles of one component to the total number of moles (solute and solvents) present in the solution. It is expressed as ‘x’.
Mole fraction of the solute = Moles of the solute / Moles of solute + Moles of solvent
Mole fraction of the solvent = Moles of the solvent / Moles of solute + Moles of solvent
Mole fraction of the solute + Mole fraction of solvent = 1
Important Points
1. Anything which has mass and occupies space is called matter.
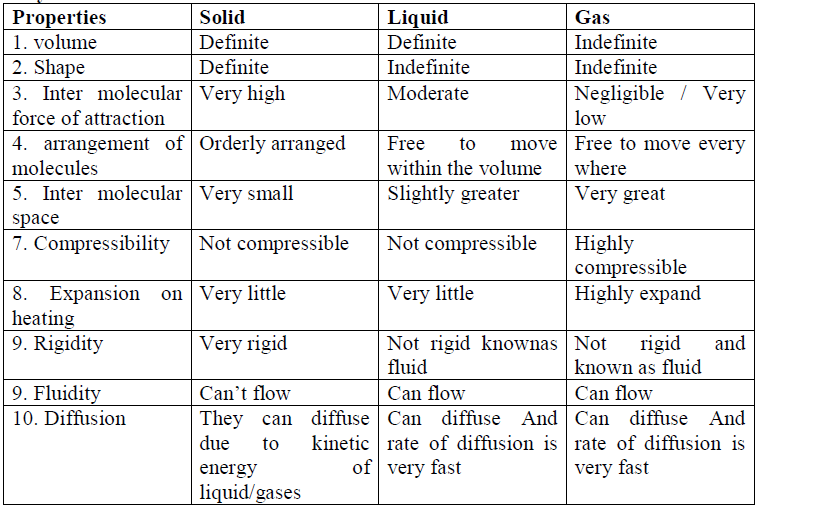
2. Matters exist in three physical states viz. solid, liquid and gas.
3. In solids, these particles are held very close to each other in an orderly fashion and there is not much freedom of movement. In liquids, the particles are close to each other but they can move around. However, in gases, the particles are far apart as compared to those present in solid or liquid states and their movement is easy and fast.
4. Solids have definite volume and definite shape.
5. Liquids have definite volume but not the definite shape. They take the shape of the container in which they are placed.
6. Gases have neither definite volume nor definite shape. They completely occupy the container in which they are placed.
7. A mixture contains two or more substances present in it (in any ratio) which are called its components.
8. A mixture may be homogeneous or heterogeneous.
9. In a homogeneous mixture, the components completely mix with each other and its composition is uniform throughout. Sugar solution and air are thus, the examples of homogeneous mixtures.
10. In heterogeneous mixtures, the composition is not uniform throughout and sometimes the different components can be observed. For example, the mixtures of salt and sugar, grains and pulses along with some dirt (often stone) pieces, are heterogeneous mixtures..
11.The components of a mixture can be separated by using physical methods such as simple hand picking, filtration, crystallization, distillation etc.
12. Pure substances have characteristics different from the mixtures. They have fixed composition, Copper, silver, gold, water, glucose are some examples of pure substances. Glucose contains carbon, hydrogen and oxygen in a fixed ratio and thus, like all other pure substances has a fixed composition. Also, the constituents of pure substances cannot be separated by simple physical methods.
13.An element consists of only one type of particles. These particles may be atoms or molecules. Sodium, copper, silver, hydrogen, oxygen etc. are some examples of elements. They all contain atoms of one type. However, the atoms of different elements are different in nature. Some elements such as sodium or copper, contain single atoms held together as their constituent particles whereas in some others, two or more atoms combine to give molecules of the element. Thus, hydrogen, nitrogen and oxygen gases consist of molecules in which two atoms combine to give their respective molecules.
14.When two or more atoms of different elements combine, the molecule of a compound is obtained. The examples of some compounds are water, ammonia, carbon dioxide, sugar etc. the atoms of different elements are present in a compound in a fixed and definite ratio and this ratio is characteristic of a particular compound.
15.The SI system(Systeme International d Unités –abbreviated as SI)) has seven base units and they are listed in Table 1.1
16. Mass of a substance is the amount of matter present in it while weight is the force exerted by gravity on an object. The mass of a substance is constant whereas its weight may vary from one place to another due to change in gravity.
17. Volume has the units of (length) 3. So in SI system, volume has units of m3. A common unit, litre (L) which is not an SI unit, is used for measurement of volume of liquids. 1 L = 1000 mL , 1000 cm3 = 1 dm3
18. Density of a substance is its amount of mass per unit volume SI units of density kg m-3
This unit is quite large and a chemist often expresses density in g cm-3
19. There are three common scales to measuretemperature — °C (degree celsius), °F (degree Fahrenheit) and K (Kelvin). Here, K is the SI unit.
20.The Kelvin scale is related to Celsius scale as follows :K =°C+273.15
21.The °F scale is related to Celsius scale as follows
°F=9/5(°C )+32
22. In scientific notation (exponential Notation) any number can be represented in the form N × 10n where n is an exponent having positive or negative values and N can vary between 1 to 10. Thus, we can write 232.508 as 2.32508 X102 in scientific notation. Note that while writing it, the decimal had to be moved to the left by two places and same is the exponent (2) of 10 in the scientific notation. Similarly, 0.00016 can be written as 1.6 X10–4. Here the decimal has to be moved four places to the right and (– 4) is the exponent in the scientific notation.
23. Significant figures are meaningful digits which are known with certainty. The uncertainty is indicated by writing the certain digits and the last uncertain digit. Thus, if we write a result as 11.2 mL, we say the 11 is certain and 2 is uncertain and the uncertainty would be ± 1 in the last digit. Unless otherwise stated, an uncertainty of +1 in the last digit is always understood.
24. There are certain rules for determining the number of significant figures. These are stated below:
a) All non-zero digits are significant. For example in 285 cm, there are three significant figures and in 0.25 mL, there are two significant figures.
b) Zeros preceding to first non-zero digit are not significant. Such zero indicates the position of decimal point. Thus, 0.03 has one significant figure and 0.0052 has two significant figures.
c) Zeros between two non-zero digits are significant. Thus, 2.005 has four significant figures.
d) Zeros at the end or right of a number are significant provided they are on the right side of the decimal point. For example, 0.200 g has three significant figures. But, if otherwise, the terminal zeros are not significant if there is no decimal point. For example, 100 has only one significant figure, but 100. has three significant figures and 100.0 has four significant figures. Such numbers are better represented in scientific notation. We can express the number 100 as 1×102 for one significant figure, 1.0×102 for two significant figures and 1.00×102 for three significant figures.
e) Counting numbers of objects, for example, 2 balls or 20 eggs, have infinite significant figures as these are exact numbers and can be represented by writing infinite number of zeros after placing a decimal i.e.,2 = 2.000000 or 20 = 20.000000
f) In numbers written in scientific notation, all digits are significant e.g., 4.01×102 has three significant figures, and 8.256 × 10-3 has four significant figures.
25. .Law of Conservation of Mass states that matter can neither be created nor destroyed.
26.Law of Definite Proportions states that a given compound always contains exactly the same proportion of elements by weight.
27.Law of Multiple Proportions states that if two elements can combine to form more than one compound, the masses of one element that combine with a fixed mass of the other element, are in the ratio of small whole numbers.
28. Gay Lussac’s Law of Gaseous Volumes: This law was given by Gay Lussac in 1808. He observed that when gases combine or are produced in a chemical reaction they do so in a simple ratio by volume provided all gases are at same temperature and pressure.
29. In 1811, Avogadro proposed that equal volumes of gases at the same temperature and pressure should contain equal number of molecules.
30. In 1808, Dalton published ‗A New System of Chemical Philosophy‘ in which he proposed the following :
a) Matter consists of indivisible atoms.
b) All the atoms of a given element have identical properties including identical mass. Atoms of different elements differ in mass.
c) Compounds are formed when atoms of different elements combine in a fixed ratio.
d) Chemical reactions involve reorganization of atoms. These are neither created nor destroyed in a chemical reaction.
e) Dalton‘s theory could explain the laws of chemical combination.
31.One atomic mass unit is defined as a mass exactly equal to one twelfth the mass of one carbon – 12 atom.
32. Molecular mass is the sum of atomic masses of the elements present in a molecule. It is obtained by multiplying the atomic mass of each element by the number of its atoms and adding them together.
33. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon-12; its symbol is ―mol.‖ When the mole is used, the elementary entities must be specified and may be atoms, molecules, ions, electrons, other particles, or specified groups of such particles. This number of entities in 1 mol is so important that it is given a separate name and symbol. It is known as ‗Avogadro constant‘, denoted by NA in honor of Amedeo Avogadro.
34.An empirical formula represents the simplest whole number ratio of various atoms present in a compound whereas the molecular formula shows the exact number of different types of atoms present in a molecule of a compound.
35.Many a time, the reactions are carried out when the reactants are not present in the amounts as required by a balanced chemical reaction. In such situations, one reactant is in excess over the other. The reactant which is present in the lesser amount gets consumed after sometime and after that no further reaction takes place whatever be the amount of the other reactant present. Hence, the reactant which gets consumed, limits the amount of product formed and is, therefore, called the limiting reagent.
36. Mass per cent = Mass of solute per 100 g of solution
=Mass of solute / Mass of solution X 100
It is the amount of solute in grams dissolved per 100 g of solution. e.g., 10% solution of sodium chloride means 10 g of solid sodium chloride present in 100 g of solution.
37. Mole Fraction: It is ratio of number of moles of a particular component to the total number of moles of all the components.
Mole-fraction of solute = No. of moles of solute / No. of moles of solute + No. of moles of solvent

38. Molality (m). It is defined as number of moles of solute (B) per 1000 g or 1 kg of solvent.
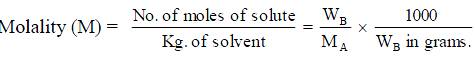
where WA is mass of solvent.
39.Molarity (M). It is expressed as the number of moles of solute per litre of solution.
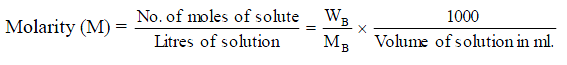
where WB is mass of solute, MB is molar mass of solute

